Market Trends of Europe Clinical Trials Industry
This section covers the major market trends shaping the Europe Clinical Trials Market according to our research experts:
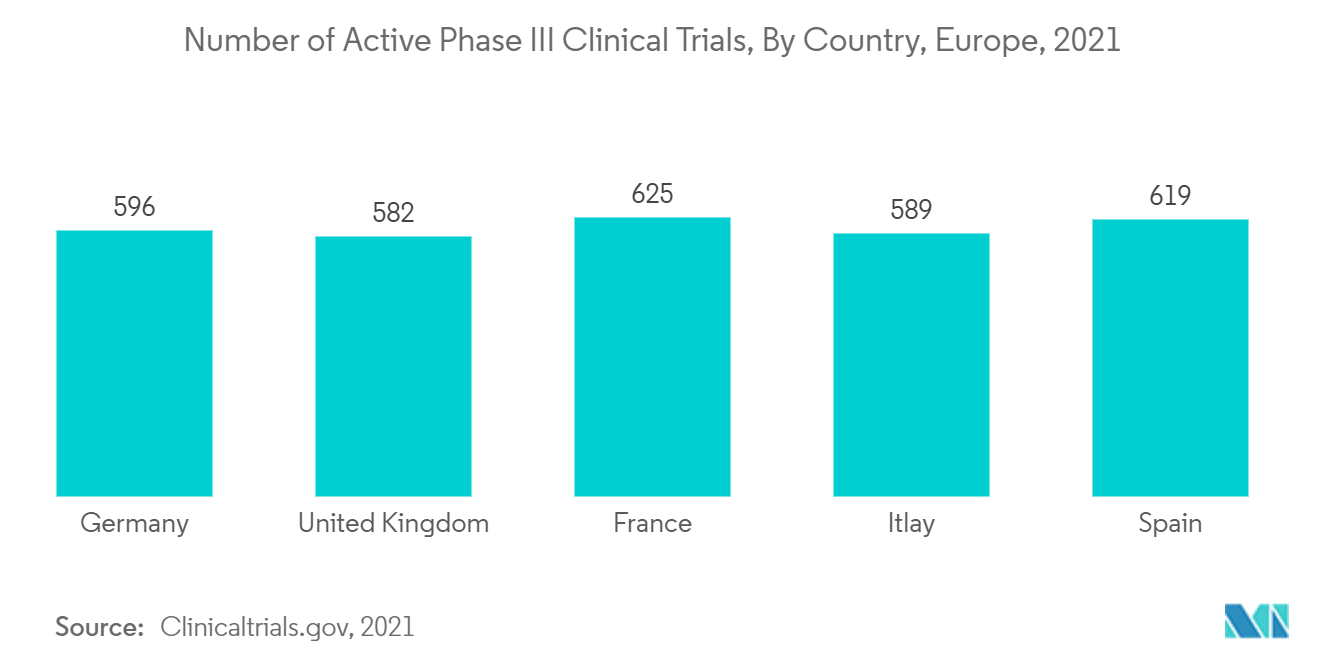
Phase III Segment is Expected to Hold the Major Revenue Share During the Forecast Period
In phase III, the drug or the treatment is tested in large groups of people, ranging from 1,000 to 3,000 participants. These trials are conducted to confirm and expand on the safety and effectiveness results from Phase I and II trials, to compare the drug to standard therapies for the disease or condition being studied, and to evaluate the overall risks and benefits of the drug. The COVID-19 pandemic has driven the growth of the market studied owing to the increasing demand for effective therapeutics. For instance, according to the European clinical trials registry in 2021, around 267 clinical trials were found to be active for COVID-19.
Phase III is one of the most critical phases in assessing the new intervention's effectiveness and its value in clinical practice. As per clinical trials.gov, in 2021, over 3,011 clinical trials in European Union were active in Phase 3 for various chronic disorders. Such a high number of clinical trials in the European Union is expected to drive the growth of the studied segment.
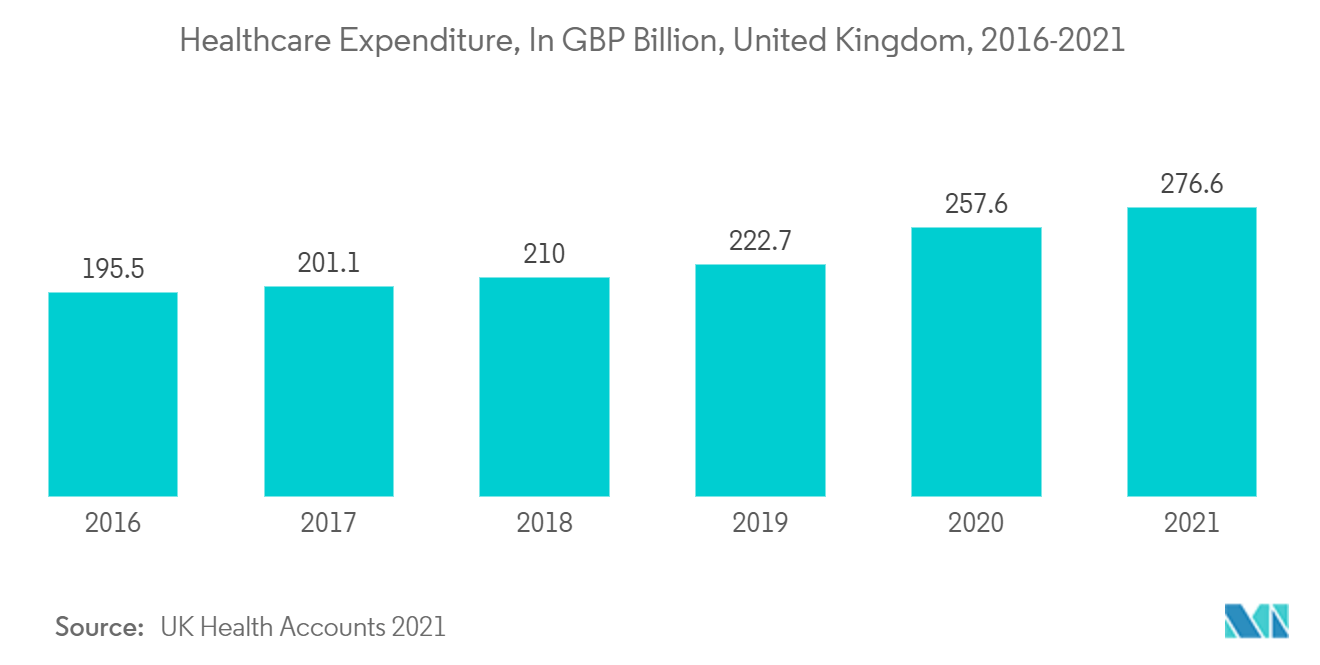
United Kingdom is Expected to Hold the Major Revenue Share During the Forecast Period
The clinical trials market is expected to witness strong growth in the United Kingdom and is anticipated to maintain the same growth trend over the forecast period. The burden of increasing chronic diseases and the increasing number of development activities necessitating clinical trials are predicted to boost the studied market growth in the United Kingdom.
There has been an increase in the prevalence of cancer cases in the United Kingdom in recent years. For instance, as per the Globocan 2020 report, over 458,000 cancer cases were reported in the year 2020 and the number of cancer cases is projected to reach 596,000 cases by the year 2040. Therefore, the rising burden of cancer is predicted to lead to an increase in development activities, creating demand for clinical trials.
Additionally, various research indicated that the development of self-injection devices such as pre-filled syringes could positively impact autoimmune diseases, for instance, rheumatoid arthritis (RA) medication adherence, and patient-centered care. As there is a burden of RA in the country, the demand for the development of drugs and drug-delivery devices is increasing, this is further garnering the demand for clinical trials. For instance, as per the 2022 update of the Nationl Rheumatoid Arthritis Society (NRAS), nearly 400,000 people in the United Kingdom had RA in 2021, which is 2 to 3 times more common in women than in men. Such a high prevalence of disease and its burden is expected to fuel the market growth in the country.
Moreover, the data published by the Birtish Heart Foundation (BHF) in August 2022 shows that around 7.6 million people were living with heart and circulatory diseases, and nearly 4 million males and 3.6 million females were living with heart and circulatory diseases in the country in 2021. Hence, the prevalence of various cardiovascular diseases is expected to drive the demand for the development of effective drugs and devices, thereby contributing to the growth of the market.
The activities and investments related to clinical trials are accelerating in the United Kingdom which is further expected to fuel the studied market growth. For instance, in November 2022, CV6 Therapeutics reported investing almost GBP 8 million (USD 9.79 million) in first-stage clinical trials for a new anti-cancer drug. Similarly, in June 2022, GSK planned to invest GBP 1 billion (USD 1.22 million) in R&D over ten years to get ahead of infectious diseases in lower-income countries. Such investments by the major players are anticipated to create numerous opportunities for the studied market growth.
Therefore, owing to the factors mentioned above, the studied market is anticipated to fuel growth over the forecast period.