Market Size of Body Contouring Devices Industry
| Study Period | 2019 - 2029 |
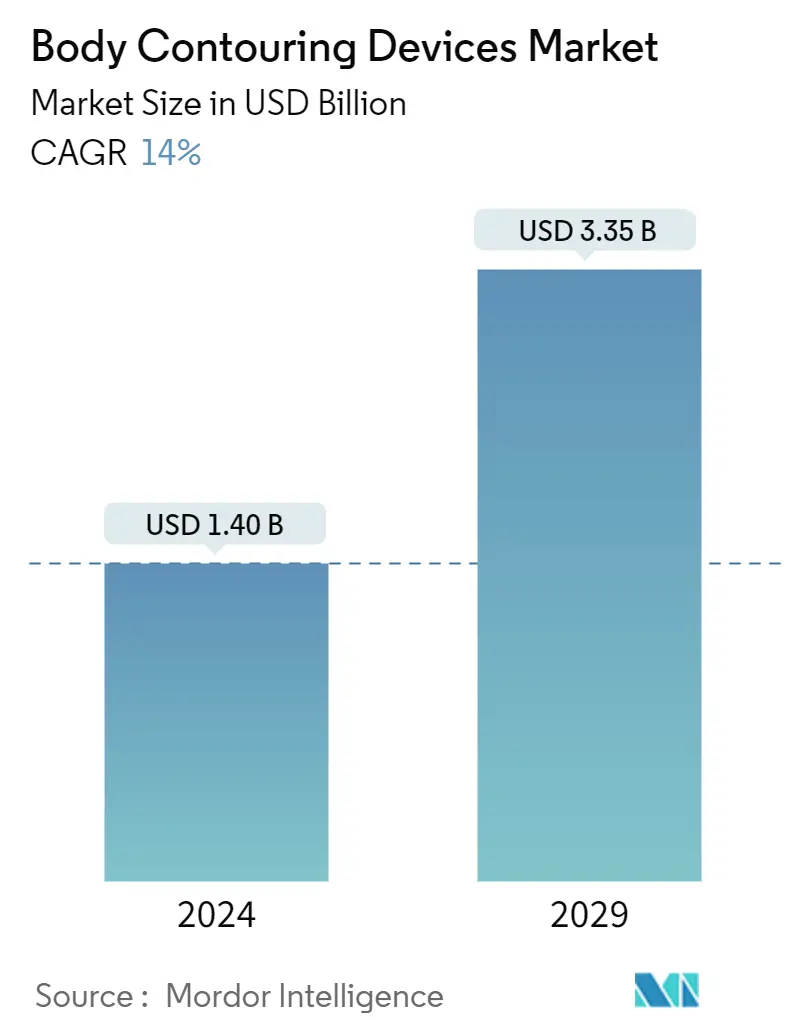
| Market Size (2024) | USD 1.40 Billion |
| Market Size (2029) | USD 3.35 Billion |
| CAGR (2024 - 2029) | 14.00 % |
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Body Contouring Devices Market Analysis
The Body Contouring Devices Market size is estimated at USD 1.40 billion in 2024, and is expected to reach USD 3.35 billion by 2029, growing at a CAGR of 14% during the forecast period (2024-2029).
The body contouring devices market is poised to register a CAGR of 17% over the forecast period.
The COVID-19 pandemic had a significant impact on the body contouring device market. For instance, an article published by Aesthetic Plastic Surgery in April 2023 observed that the motivation level of the participants who underwent facial, body contouring, and rhinoplasty surgery for 'bothered by their appearance in the mirror' was statistically significantly lower during the pandemic period compared to the pre-pandemic period. However, as the market returns to its pre-pandemic state due to the decline in COVID-19 cases and increased concern about body aesthetics, it will show significant growth over the forecast period.
The growing adoption of minimally invasive body contouring procedures and advancements in device technologies are the significant factors boosting the growth of the body contouring devices market. For instance, according to the World Health Organization Report in March 2022, over 1 billion individuals, including 650 million adults, 340 million teenagers, and 39 million children, are obese worldwide. Additionally, 167 million adults and children will have worse health by 2025 as a result of being overweight or obese. Thus, with the increase in the obese population, the demand for body contouring is expected to increase over the forecast period.
Furthermore, the companies are actively involved in new product launch developments and collaborations to expand their footprint. For instance, in April 2023, Apex Medical Corporation received 510(k) clearance from the U.S. FDA for using the Renuvion APR Handpiece "for coagulation of subcutaneous soft tissues following liposuction for aesthetic body contouring." It demonstrated the safety and effectiveness of Renuvion technology and continued commitment to working with the FDA to secure specific clinical indications.
Thus, all factors above are expected to boost market growth over the forecast period. However, body contouring devices are expensive, and there is a lack of awareness of them in many parts of the world.
