Market Size of Asia Pacific In Vitro Diagnostics Industry
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
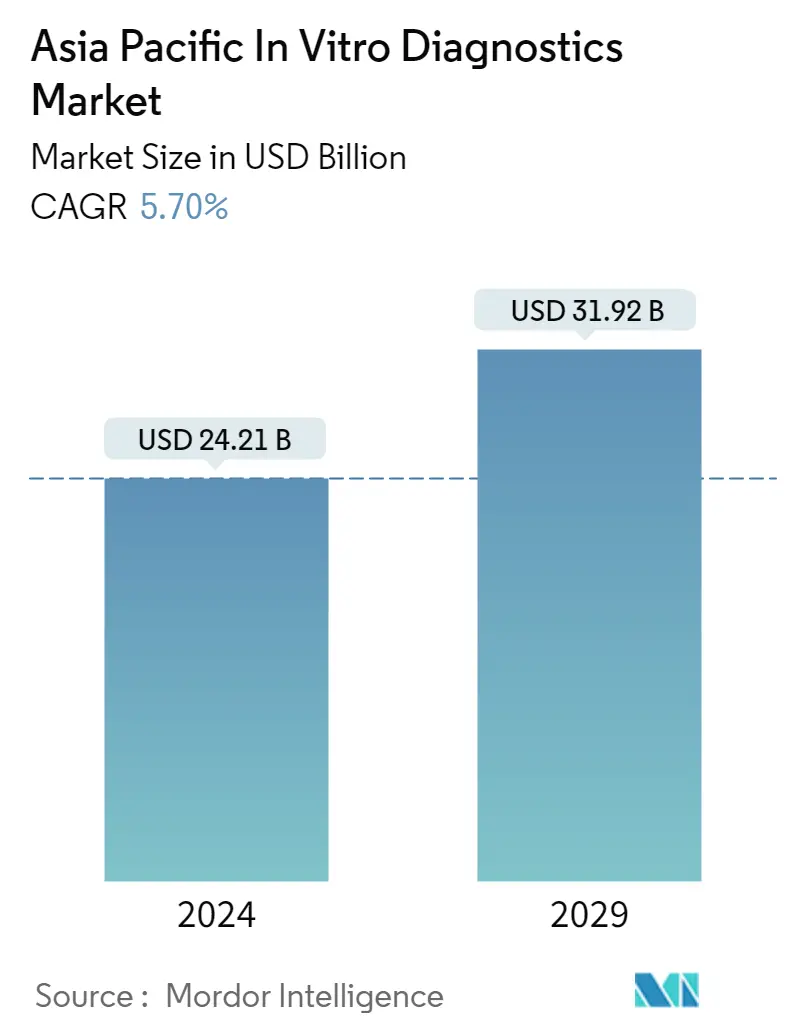
| Market Size (2024) | USD 24.21 Billion |
| Market Size (2029) | USD 31.92 Billion |
| CAGR (2024 - 2029) | 5.70 % |
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Asia Pacific In-Vitro Diagnostics Market Analysis
The Asia Pacific In Vitro Diagnostics Market size is estimated at USD 24.21 billion in 2024, and is expected to reach USD 31.92 billion by 2029, growing at a CAGR of 5.70% during the forecast period (2024-2029).
The COVID-19 outbreak affected the Asia-Pacific in-vitro diagnostic market's growth adversely in its preliminary phase. However, this market gained traction due to the significant urge and rapid demand for newly launched emergency-use authorized in-vitro diagnostic tests for COVID-19 detection. For instance, in May 2022, Cipla Limited started the commercialization of the 'RT-Direct' multiplex COVID-19 RT-PCR Test kit in India in partnership with Genes2Me Pvt. Ltd. This kit was validated at an Indian Council of Medical Research (ICMR)-approved center. However, the rapid demand for newly launched emergency-use authorized in-vitro diagnostic tests for COVID-19 detection compensated for this detrimental impact. However, the market is growing at a stable pace due to a resumption of diagnosis procedures, manufacturing activities, and availability of products. It is expected to witness a similar trend over the coming years.
Further, the high cases of chronic diseases, the increasing use of point-of-care (POC) diagnostics, advanced technologies in vitro diagnostic products, and increasing awareness and use of personalized medicine and companion diagnostics are among the major factors driving the growth of the studied market. For instance, the 2021 International Diabetes Federation (IDF) projects that the number of people with diabetes in Southeast Asia will increase by 68%, reaching 152 million by 2045. The same report also reported that the Western Pacific region holds the third-highest prevalence of diabetes (11.9%) globally. The number of diabetic cases is estimated to increase in the Western Pacific region by 27%, reaching 260 million by 2045, and the prevalence of diabetes will increase by 21% in 2045. Such a high incidence of chronic diseases like diabetes in the given region is increasing the demand for diagnostics associated with the diseases.
Additionally, as per the article published in the Chinese Medical Journal in March 2022, China is experiencing a greater cancer incidence. In China, there were about 4.8 million new cancer cases in 2022, with lung cancer being the most prevalent type. Thus, the increasing incidence of cancer cases increased the demand for associated diagnostics, contributing to the market.
The strategies adopted by the market players, such as product launches, partnerships, and collaboration, are expected to propel the market's growth. For instance, in March 2021, OpGen announced that it had received regulatory approval from the Chinese National Medical Products Administration for its Curetis Unyvero System as an in-vitro diagnostics instrument. In April 2021, Otsuka Pharmaceutical Co., Ltd., in collaboration with Denka Company Limited, launched the Helicobacter Pylori test kit, QuickNavi-H. Pylori. It is a diagnostic kit that utilizes immunochromatography to confirm the presence or absence of Helicobacter Pylori antigens in a stool sample. The test kit will be used in medical institutions as it enables rapid evaluation in eight minutes by adding three drops of the sample liquid to the kit. Thus, the market can show significant growth over the forecast period due to these technological advances.
Thus, the reasons mentioned above may drive the market. However, stringent regulations regarding product approvals and cumbersome reimbursement procedures may slow the market's growth.
