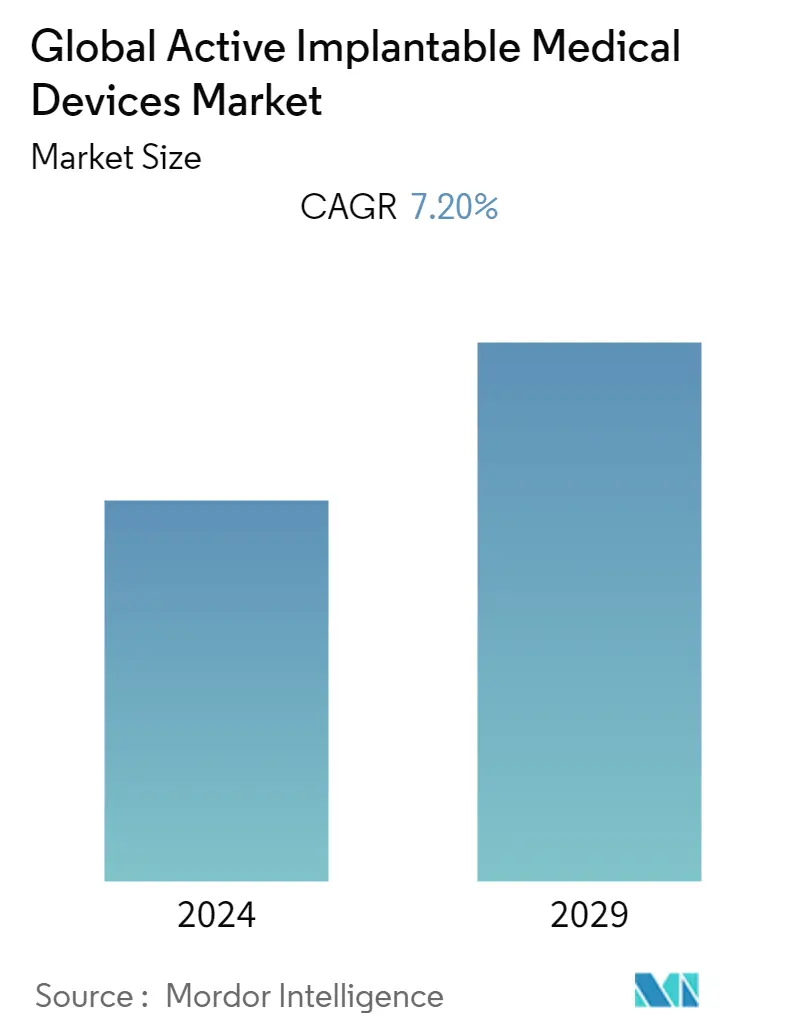
Market Size of Global Active Implantable Medical Devices Industry
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
| CAGR | 7.20 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Active Implantable Medical Devices Market Analysis
The active implantable medical devices market is projected to register a CAGR of 7.2% during the forecast period.
The COVID-19 pandemic has had a large impact on the creation and use of medical devices, as well as most aspects of healthcare. Patients with cardiac implantable electronic devices (CIED) declined scheduled follow-up appointments during the global COVID-19 pandemic due to the risk of infection. Remote monitoring is intended to optimize the clinical treatment of CIED patients, enhance their quality of life, and decrease access to emergency rooms and hospitals. As per an article titled 'Implantation of cardiac electronic devices in active COVID-19 patients: Results from an international survey' published in February 2022, in hospitals around the world, Cardiac implantable electronic device (CIED) procedure rates for known active COVID-19 disease ranged from 0 to 16.2 per 1000 patients. High rates of complications and mortality were observed in patients with active COVID-19 infection who had CIED implantation. Before performing CIED implantation in patients with active COVID-19, operators should weigh these risks. These abovementioned factors are expected to have a significant impact on the growth of the market growth during and after the post-pandemic.
Certain factors that are driving the market growth include the increasing burden of cardiovascular and neurological diseases and investments and funds to develop technologically advanced products. As per an article titled 'The prevalence and predictors of cardiovascular diseases in Kherameh cohort study: a population-based study on 10,663 people in southern Iran' published in, the prevalence of Cardiovascular Disease (CVD) increased from 257 million in 1990 to 550 million in 2020. Evidence has also indicated the global prevalence of angina to be 0.73-14.4% in females and 0.76-15.1% amongst males. The market for implantable devices is expected to grow due to several factors, including an aging population, an increase in cardiovascular and neurological diseases, investments in developing technologically advanced products, expanded uses for neurostimulators, and favorable reimbursement conditions for ENT procedures in developed nations.
Furthermore, increasing chronic diseases coupled with the advancements in active implantable medical devices are projected to boost market growth. As per an article titled 'Commentary on Chronic Disease Prevention in 2022' published in 2022, nearly 60% of adult Americans have at least one chronic disease. Chronic conditions like diabetes, cancer, and cardiovascular disease are the leading causes of death in the United States.
Product approvals and launches are expected to boost the market growth. For instance, in December 2021, LifeTech Scientific Corporation extended its agreements with Medtronic to further the strategic collaboration on the "HeartTone domestic pacemaker project" and to start the collaboration on domestically-made MRI-conditional pacemakers. Additional to that, in February 2021, Boston Scientific Corporation initiated the MODULAR ATP clinical trial to evaluate the safety, performance and effectiveness of the mCRM Modular Therapy System. The mCRM system consists of two cardiac rhythm management (CRM) devices intended to work together to coordinate therapy: the EMBLEM MRI subcutaneous implantable defibrillator system and the empower modular pacing system (MPS), which is designed to be the one of the first leadless pacemaker capable of delivering both bradycardia pacing support and antitachycardia pacing (ATP).
However, rising demand for non-invasive procedures, issues related to implantable devices, and the high cost of the devices are anticipated to impede market growth.
