Market Size of Antinuclear Antibody Test Industry
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
| CAGR | 12.50 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Antinuclear Antibody Test Market Analysis
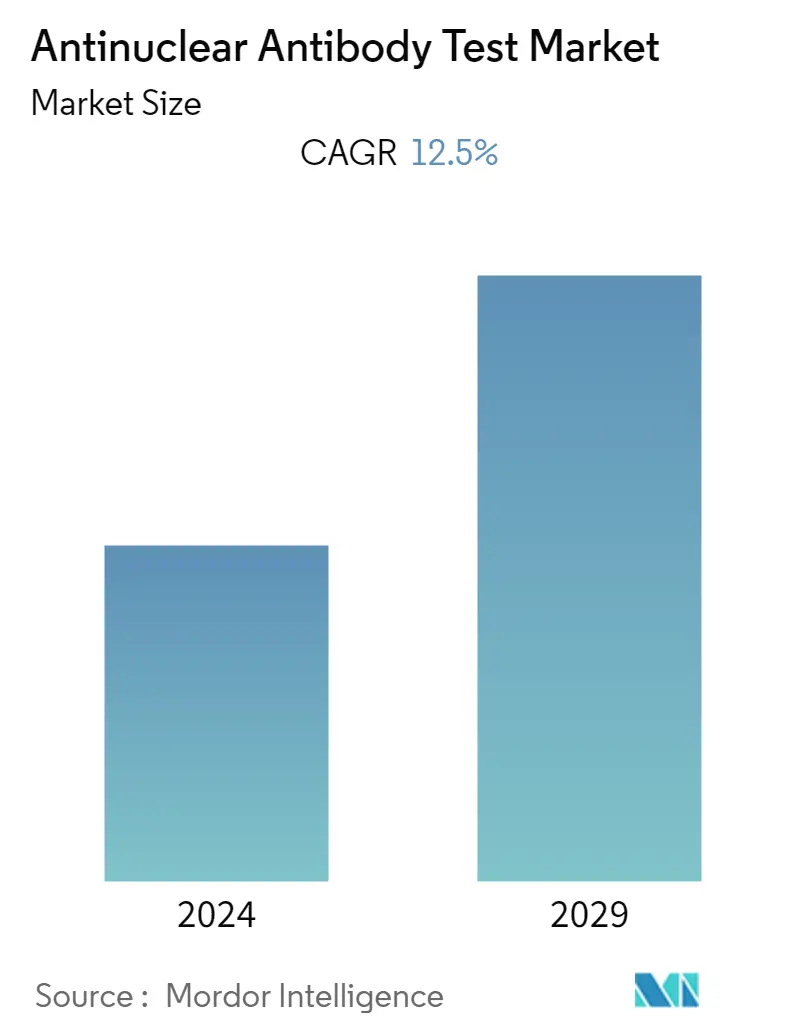
The antinuclear antibody test (ANA) market studied was anticipated to grow with a CAGR of 12.5% over the forecast period.
COVID-19 had a significant impact on the growth of the market during the forecast period. The study was conducted where a team of researchers screened several systemic autoantibodies to assess their association with the severity of COVID-19. The results were published in the article of medRxiv in January 2022, which stated that antinuclear antibodies (ANA) would be prevalent in COVID-19 patients up to one year after recovering from the SARS-CoV-2 infection. As a result, there is a good chance that the need for autoantibody diagnosis in COVID-19-infected patients will rise. This diagnosis may be done using an antinuclear antibody test. However, the number of COVID-19 cases has been subsidized but the risk of spread of infection due to new variants still persists and as the antinuclear antibody test is used in such diagnosis , the market is anticipated to have stable growth over the next 2-3 years.
Factors such as rising government initiatives with growing incidences of autoimmune diseases and favorable medical insurance and reimbursement policies are expected to drive market growth. Additionally, rising product launches will also boost the market. The increasing prevalence of autoimmune disorders, such as Sjogren's syndrome, acute motor axonal neuropathy, and others, increased the adoption of ANA testing. As per NCBI updated in July 2022, Guillain-Barre syndrome (GBS), which has an incidence of 0.4 to 2 per 100,000, has a significant impact on the healthcare system. Therefore, the increasing burden associated with GBS drives the demand for its effective diagnostic assay, thereby driving the antinuclear antibody test market over the study period.
Moreover, rising research and development activities proving the efficacies of diagnostic assays will further increase their adoption and is expected to have significant growth in the market. For instance, according to a study published in March 2022 in the journal Hematology, Transfusion and Cell Therapy, every patient must have a baseline level of the fluorescent antinuclear antibody (FANA) when they are diagnosed with autoimmune hemolytic anemia (AIHA). The presence of FANA gives a prognostic value and helps in deciding the treatment options which is expected to have a positive impact on the market.
Thus, the above-mentioned factors like the increasing burden of autoimmune diseases and new developments in the market are likely to increase the demand for the ANA test and the studied market is expected to grow significantly during the study period. However, increasing concerns about the reliability of antinuclear antibody tests and lack of awareness of the disease and effective treatment in developing economies are expected to hinder the market growth.
