USA Neurology Devices Market Size
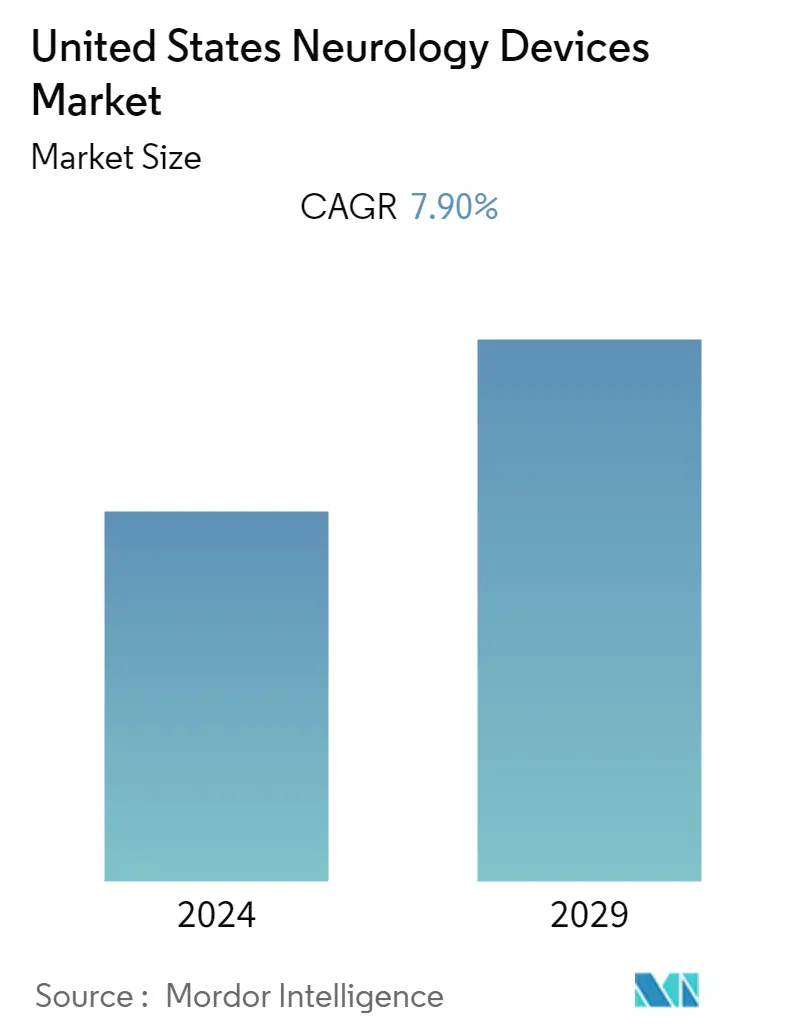
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
| Historical Data Period | 2019 - 2022 |
| CAGR | 7.90 % |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
USA Neurology Devices Market Analysis
The United States Neurology Devices Market is expected to grow with a CAGR of 7.9% in the forecast period. The key factors propelling the growth of the market are the increasing incidence of neurological disorders, huge investments by private players in neurology devices, an increase in R&D in the field of neuro therapies, and the rising aging population. The increasing incidence of neurological disorders like brain aneurysms, brain tumors, epilepsy, memory disorders, multiple sclerosis, Parkinson's disease, peripheral neuropathy, post-herpetic neuralgia, spinal cord tumor, and stroke. Furthermore, increasing healthcare spending, increasing foreign direct investment by global medical device manufacturers are boosting the market growth. However, the high cost of the devices and stringent FDA validation and guidelines for new devices is restraining market growth.
USA Neurology Devices Market Trends
This section covers the major market trends shaping the USA Neurology Devices Market according to our research experts:
Neurostimulation Devices is Expected to Show Good Growth Over the Forecast Period
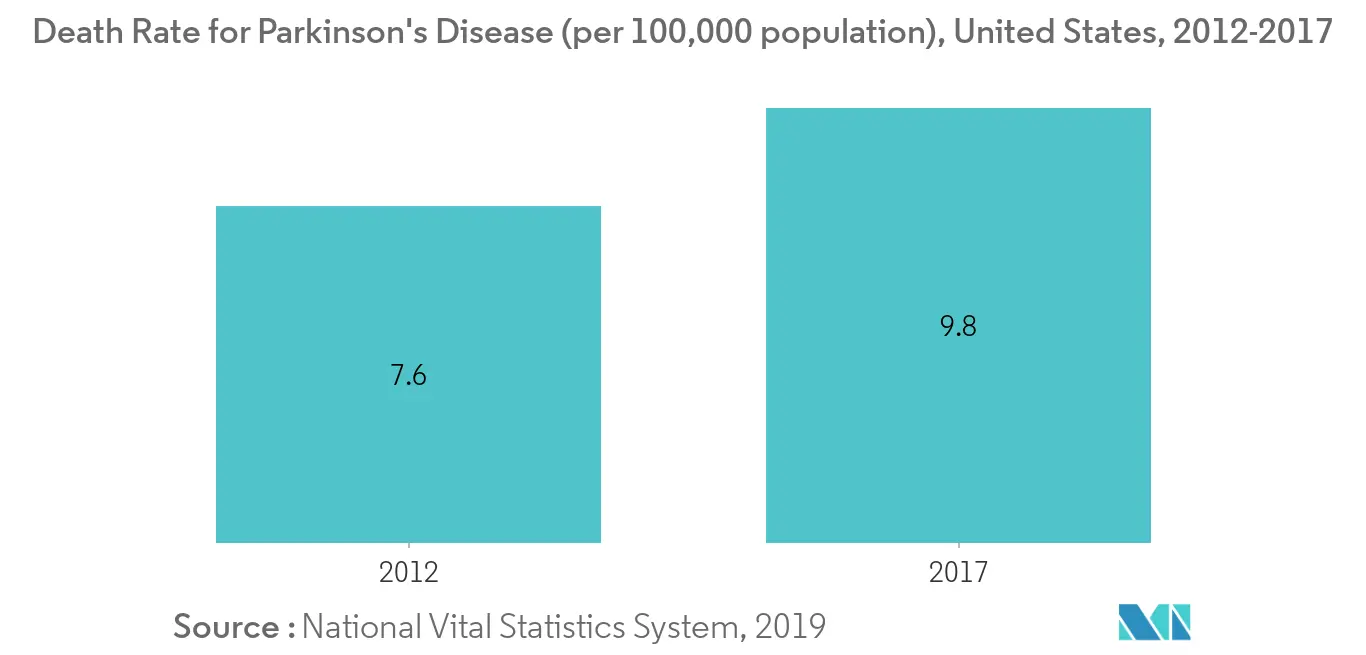
The fastest growth of the segment is due to the increasing usage of the sacral nerve stimulation devices and deep brain stimulation devices. The sacral nerve stimulation devices are mostly used for patients for whom drug therapy or other types of medications are not successful. These devices are found to be successful in treating bladder problems. Hence, with increasing incidences of overactive bladder, the sacral nerve stimulation devices segment is expected to grow during the forecast period. The deep brain stimulation devices are generally used to treat neuropsychiatric disorders and are effectively used in treating various other neurological disorders, like Parkinson's disease, essential tremor, dystonia, chronic pain, major depression, and obsessive-compulsive disorder. According to ParkinsonsDisease.net, 2019, 1 million Americans are living with PD, and approximately 60,000 Americans are diagnosed with PD each year. With the increasing neurological diseases like this, the segment is bound to grow at a higher growth rate.
USA Neurology Devices Industry Overview
The United States Neurology Devices Market is moderately competitive with several major players. Some of the strategies implemented include agreements, collaborative models, business expansion, and product developments. Some of the major players are B. Braun Melsungen AG, Boston Scientific Corporation, Stryker Corporation, Medtronic PLC, Abbott Laboratories, Johnson and Johnson, Smith & Nephew, etc.
USA Neurology Devices Market Leaders
B. Braun Melsungen AG
Boston Scientific Corporation
Stryker Corporation
Medtronic PLC
Abbott Laboratories
*Disclaimer: Major Players sorted in no particular order
USA Neurology Devices Market Report - Table of Contents
-
1. INTRODUCTION
-
1.1 Study Deliverables
-
1.2 Study Assumptions
-
1.3 Scope of the Study
-
-
2. RESEARCH METHODOLOGY
-
3. EXECUTIVE SUMMARY
-
4. MARKET DYNAMICS
-
4.1 Market Overview
-
4.2 Market Drivers
-
4.2.1 Increase Burden of Neurological Disorders
-
4.2.2 Huge Investments by Private Players in Neurology Devices
-
4.2.3 Increase in R&D in the Field of Neurotherapies
-
-
4.3 Market Restraints
-
4.3.1 High Cost of Equipment
-
4.3.2 Stringent FDA Regulatory Guidelines for New Devices
-
-
4.4 Porter's Five Force Analysis
-
4.4.1 Threat of New Entrants
-
4.4.2 Bargaining Power of Buyers/Consumers
-
4.4.3 Bargaining Power of Suppliers
-
4.4.4 Threat of Substitute Products
-
4.4.5 Intensity of Competitive Rivalry
-
-
-
5. MARKET SEGMENTATION
-
5.1 By Type of Device
-
5.1.1 Cerebrospinal Fluid Management Devices
-
5.1.2 Interventional Neurology Devices
-
5.1.2.1 Interventional/Surgical Simulators
-
5.1.2.2 Neurothrombectomy Devices
-
5.1.2.3 Carotid Artery Stents
-
5.1.2.4 Embolic Coils
-
5.1.2.5 Support Devices
-
-
5.1.3 Neurosurgery Devices
-
5.1.3.1 Neuroendoscopes
-
5.1.3.2 Stereotactic Systems
-
5.1.3.3 Aneurysm Clips
-
5.1.3.4 Other Neurosurgery Devices
-
-
5.1.4 Neurostimulation Devices
-
5.1.4.1 Spinal Cord Stimulation Devices
-
5.1.4.2 Deep Brain Stimulation Devices
-
5.1.4.3 Sacral Nerve Stimulation Devices
-
5.1.4.4 Other Neurostimulation Devices
-
-
5.1.5 Other Types of Devices
-
-
-
6. COMPETITIVE LANDSCAPE
-
6.1 Company Profiles
-
6.1.1 B. Braun Melsungen AG
-
6.1.2 Boston Scientific Corporation
-
6.1.3 Stryker Corporation
-
6.1.4 Medtronic PLC
-
6.1.5 Abbott Laboratories
-
6.1.6 Johnson and Johnson
-
6.1.7 Smith & Nephew
-
6.1.8 MicroPort Scientific Corporation
-
6.1.9 Nihon Kohden Corporation
-
6.1.10 Penumbra Inc.
-
- *List Not Exhaustive
-
-
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
USA Neurology Devices Industry Segmentation
As per the scope of the report, neurological devices are medical devices that help to diagnose, prevent, and treat a variety of neurological disorders and conditions, such as Alzheimer's disease, Parkinson's disease, major depression, and traumatic brain injury.
| By Type of Device | |||||||
| Cerebrospinal Fluid Management Devices | |||||||
| |||||||
| |||||||
| |||||||
| Other Types of Devices |
USA Neurology Devices Market Research FAQs
What is the current United States Neurology Devices Market size?
The United States Neurology Devices Market is projected to register a CAGR of 7.90% during the forecast period (2024-2029)
Who are the key players in United States Neurology Devices Market?
B. Braun Melsungen AG, Boston Scientific Corporation, Stryker Corporation, Medtronic PLC and Abbott Laboratories are the major companies operating in the United States Neurology Devices Market.
What years does this United States Neurology Devices Market cover?
The report covers the United States Neurology Devices Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the United States Neurology Devices Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
United States Neurology Devices Industry Report
Statistics for the 2024 United States Neurology Devices market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. United States Neurology Devices analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
