Hepatitis Therapeutics Market Size
| Study Period | 2021 - 2029 |
| Base Year For Estimation | 2023 |
| Forecast Data Period | 2024 - 2029 |
| CAGR | 3.50 % |
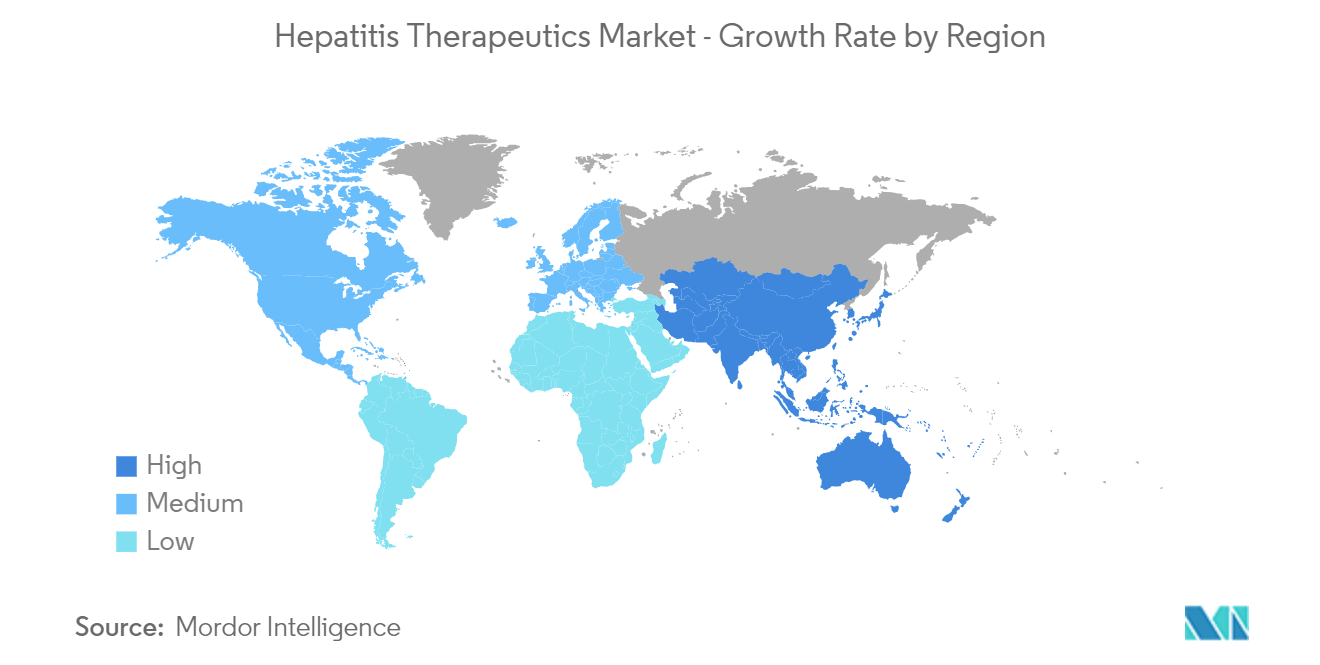
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Hepatitis Therapeutics Market Analysis
During the time frame of the forecast, the hepatitis therapeutics market is expected to grow at a CAGR of 3.5%.
The impact of COVID-19 on healthcare services had a substantial impact on the delivery of care and outcomes for patients with chronic diseases. Therefore, there was a decline in the treatment, diagnosis, and follow-up of patients with hepatitis B and C, which had a significant impact on the healthcare industry. Additionally, clinical trials of the drugs were reduced as more and more companies delayed the start of most new courses in several countries. In addition, the COVID-19 epidemic saw a decline in outpatient services for patients with chronic hepatitis, which also had a significant impact on overall market growth. The study published in July 2021 in PubMed stated that the COVID-19 pandemic affected hepatitis prevention, testing, treatment, and vaccination services globally. But the return of elective care procedures around the world is likely to bring the market's growth back to where it was before the pandemic.
The market is anticipated to witness rapid growth due to the growing incidence of hepatitis, growing government support for awareness of hepatitis diseases, and increased availability of highly technologically advanced medical products. Hepatitis is an inflammation of the liver. Hepatitis may be due to drugs, alcohol consumption, or other medical conditions affecting the liver. But in most cases, it is caused by a virus called viral hepatitis, and the most common types are hepatitis A, B, and C. Hepatitis B is very common and can lead to severe cirrhosis of the liver, which can be fatal. Moreover, government initiatives to increase awareness regarding hepatitis and its treatment are anticipated to boost market growth over the forecast period. For instance, in June 2022, the "World Hepatitis Summit (WHS)" occurred as a virtual event. The World Hepatitis Summit 2022 was co-sponsored by the World Health Organization (WHO). The summit provides a platform for a broad hepatitis community to take stock of progress to date and share ideas, experiences, and best practises in addressing the many challenges of viral hepatitis.
Furthermore, according to the Hepatitis B Foundation 2022 update, two billion people have been infected with the hepatitis B virus (one out of three people). Approximately 1.5 million people become newly infected each year. Almost 300 million people are chronically infected. Thus, the increasing burden of hepatitis is propelling the growth of the market. Additionally, in March 2021, Dicerna Pharmaceuticals and Roche commenced the Phase II combination trial of the investigational GalXC RNAi candidate, RG6346, for the treatment of chronic hepatitis B virus (HBV) infection. In addition, in March 2022, VBI Vaccines Inc. received FDA approval for the 3-antigen HBV vaccine for adults in the United States, PreHevbrio for hepatitis B. Therefore, such research and development activities in the market are expected to drive market growth.
Therefore, the increase in the prevalence of the hepatitis B virus, the promising pipeline for hepatitis drugs, and government-sponsored programmes to encourage patients to accept hepatitis drugs are likely to drive the hepatitis therapeutics market over the forecast period. During the analysis period, the market is likely to grow slowly because of strict regulations and bad reimbursement policies.
Hepatitis Therapeutics Market Trends
This section covers the major market trends shaping the Hepatitis Therapeutics Market according to our research experts:
Hepatitis C Segment is Expected to Hold a Major Market Share in the Hepatitis Therapeutics Market
Hepatitis C is a liver disease caused by the hepatitis C virus. Hepatitis C can range in severity from mild to fatal.Acute hepatitis C occurs within the first six months after exposure to the hepatitis C virus, and chronic hepatitis C can develop into lifelong hepatitis C if left untreated. According to data published by the World Health Organization (WHO) in July 2021, an estimated 58 million people have chronic hepatitis C virus, and about 1.5 million new infections occur annually. According to the same source, the hepatitis C virus is a blood-borne pathogen, and the most common form of infection is exposure to small amounts of blood. This often happens with unsafe injections and unsafe health care.
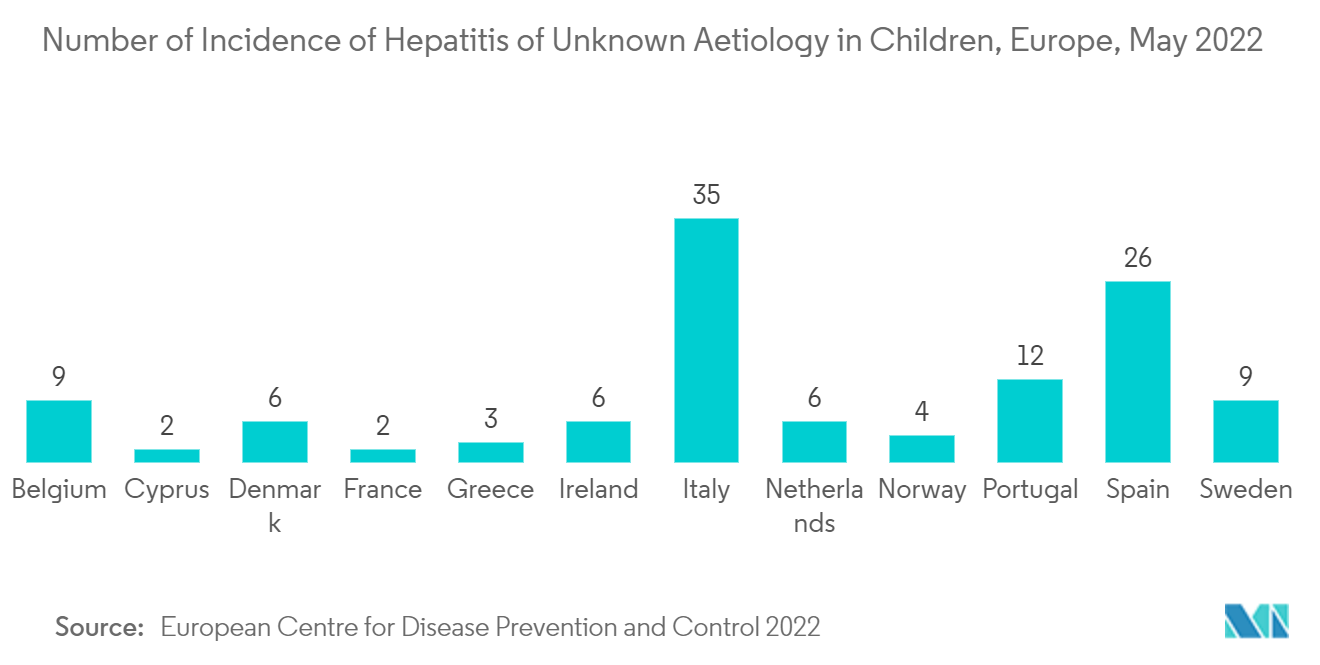
The hepatitis C segment is anticipated to witness growth in the hepatitis therapeutics market due to the rise in hepatitis infection globally. For instance, as per the ECDC 2022 report, 26 cases of hepatitis of unknown etiology were reported in May 2022 in Spain. Moreover, it is expected to show a similar trend over the forecast period due to the high prevalence of hepatitis C in developing and developed regions, as well as the growing availability of effective drugs in the market. In addition, the introduction of several products by major players is expected to boost the segment's growth. For example, in June 2021, Gilead Sciences, Inc. received approval from the FDA for Epclusa (sofosbuvir/velpatasvir) for the treatment of chronic hepatitis C virus (HCV) in children under 3 years of age, regardless of the HCV genotype or severity of the liver disease. Furthermore, the rise in clinical studies related to hepatitis C is anticipated to drive market growth. For instance, in February 2022, the Task Force for Global Health sponsored a clinical trial to assess the outcomes of hepatitis C-affected mother-infant pairs exposed to direct-acting antivirals (DAAs) during pregnancy within a global clinical case registry.
Therefore, due to the rise in the prevalence of hepatitis and the increase in product launches and clinical studies associated with the treatment of hepatitis C, the segment is anticipated to witness growth in the market over the forecast period.
North America is Expected to Hold a Significant Share in the Market and Expected to do Same in the Forecast Period
Due to an increase in hepatitis infections and an increase in the commercial availability of hepatitis drugs in the region, North America is expected to hold a significant market share in the hepatitis therapeutics market. For example, between April and September 2022, PHAC looked into 28 cases of acute severe hepatitis in children in Canada that met the national case definition.
Moreover, the data published in the report Viral Hepatitis National Strategic Plan: 2021-2025 for the United States stated that the goal of the strategic plan is to improve viral hepatitis-related health outcomes of people with viral hepatitis, reduce viral hepatitis-related disparities and health inequities, and improve viral hepatitis surveillance and data usage, etc. In addition, favorable reimbursement policies, high rates of improved access to healthcare, and the availability of well-functioning healthcare infrastructure also stimulate growth across the regional market on a large scale.
Furthermore, the presence of key market players, coupled with growing awareness programmes and the introduction of the latest products in the region, is responsible for bolstering the market's growth in the United States. For instance, in December 2021, the United States Food and Drug Administration approved Pre Hebrio [a recombinant vaccine] to prevent infections caused by all known minor hepatitis B virus (HBV) infections in adults. However, a national strategic plan was launched by the United States Department of Health and Human Services in January 2021 to eradicate hepatitis. It included five main goals, objectives and strategies to achieve these goals, and indicators with measurable objectives for monitoring progress. Therefore, the introduction of such programmes by the government will improve the treatment of hepatitis in the country.
Thus, due to the rise in the prevalence of hepatitis, the increase in government initiatives and strategic plans for hepatitis, and the surge in product launches, it is anticipated that North America will witness growth in the market over the forecast period.
Hepatitis Therapeutics Industry Overview
The hepatitis therapeutics market is competitive and consists of several major players. However, in terms of market share, only a few of the major players are currently dominating the market. Some of the leading market players include Merck & Co., Inc., Gilead Sciences, Inc., AbbVie Inc., Bristol-Myers Squibb Company, F. Hoffmann-La Roche, Ltd., LAURUS Labs, Zydus Cadila, Hetero Healthcare Limited, NATCO Pharma Limited, and Cipla Inc.
Hepatitis Therapeutics Market Leaders
AbbVie Inc.
Bristol Myers Squibb Company
F Hoffmann-La Roche Ltd
Gilead Sciences Inc.
Merck KGaA
*Disclaimer: Major Players sorted in no particular order
Hepatitis Therapeutics Market News
- November 2022: Gilead Sciences, Inc. received U.S. Food and Drug Administration (FDA) approval for the supplemental new drug application (sNDA) for Vemlidy (tenofovir alafenamide) 25 mg tablets as a once-daily treatment for chronic hepatitis B virus (HBV) infection in pediatric patients 12 years of age and older with compensated liver disease.
- April 2022: Lupin received approval from the United States Food and Drug Administration for tenofovir alafenamide tablets to treat chronic hepatitis B virus infection.
Hepatitis Therapeutics Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increasing Prevalence of Viral Hepatitis
4.2.2 Favorable Government Support for Creating Awareness about Hepatitis
4.2.3 Increasing Availability of Technologically Advanced Therapeutic Products
4.3 Market Restraints
4.3.1 Stringent Regulatory Issues
4.3.2 Poor Reimbursement Policies
4.4 Industry Attractiveness - Porter's Five Forces Analysis
4.4.1 Bargaining Power of Buyers/Consumers
4.4.2 Bargaining Power of Suppliers
4.4.3 Threat of New Entrants
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD Million)
5.1 By Disease Type
5.1.1 Hepatitis A
5.1.2 Hepatitis B
5.1.3 Hepatitis C
5.1.4 Hepatitis D
5.1.5 Other Types
5.2 By Drug Class
5.2.1 Interferon
5.2.2 Monoclonal Antibody
5.2.3 Non-structural protein 5A (NS5A) Inhibitors
5.2.4 Nucleotide Analog Reverse Transcriptase Inhibitors
5.2.5 Nucleotide Analog NS5B Polymerase Inhibitors
5.2.6 Multi Class Combination
5.2.7 Other Drug Classes
5.3 Geography
5.3.1 North America
5.3.1.1 United States
5.3.1.2 Canada
5.3.1.3 Mexico
5.3.2 Europe
5.3.2.1 Germany
5.3.2.2 United Kingdom
5.3.2.3 France
5.3.2.4 Italy
5.3.2.5 Spain
5.3.2.6 Rest of Europe
5.3.3 Asia-Pacific
5.3.3.1 China
5.3.3.2 Japan
5.3.3.3 India
5.3.3.4 Australia
5.3.3.5 South Korea
5.3.3.6 Rest of Asia-Pacific
5.3.4 Middle East and Africa
5.3.4.1 GCC
5.3.4.2 South Africa
5.3.4.3 Rest of Middle East and Africa
5.3.5 South America
5.3.5.1 Brazil
5.3.5.2 Argentina
5.3.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Merck KGaA
6.1.2 Gilead Sciences Inc.
6.1.3 AbbVie Inc.
6.1.4 Bristol Myers Squibb Company
6.1.5 F Hoffmann-La Roche, Ltd
6.1.6 LAURUS Labs
6.1.7 Zydus Cadila
6.1.8 Hetero Healthcare Limited
6.1.9 NATCO Pharma Limited
6.1.10 Cipla Inc.
6.1.11 Johnson & Johnson
6.1.12 Biocon
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Hepatitis Therapeutics Industry Segmentation
As per the scope of the report, hepatitis is an inflammation of the liver. The standard treatment for treating hepatitis includes several antiviral medications for different types of hepatitis, namely A, B, and C. The most effective therapy for combating the condition is a combination of different drugs. The Hepatitis Therapeutics Market is classified into Disease Type (Hepatitis A, B, C, D, and Others), Drug Class (Interferon, Monoclonal Antibody, Non-structural Protein 5A (NS5A) Inhibitors, Nucleotide Analog Reverse Transcriptase Inhibitors, Nucleotide Analog NS5B Polymerase Inhibitors, Multi-Class Combination, and Others), and Geography (North America, Europe, The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers the value (in USD million) for the above segments.
| By Disease Type | |
| Hepatitis A | |
| Hepatitis B | |
| Hepatitis C | |
| Hepatitis D | |
| Other Types |
| By Drug Class | |
| Interferon | |
| Monoclonal Antibody | |
| Non-structural protein 5A (NS5A) Inhibitors | |
| Nucleotide Analog Reverse Transcriptase Inhibitors | |
| Nucleotide Analog NS5B Polymerase Inhibitors | |
| Multi Class Combination | |
| Other Drug Classes |
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Hepatitis Therapeutics Market Research FAQs
What is the current Hepatitis Therapeutics Market size?
The Hepatitis Therapeutics Market is projected to register a CAGR of 3.5% during the forecast period (2024-2029)
Who are the key players in Hepatitis Therapeutics Market?
AbbVie Inc., Bristol Myers Squibb Company, F Hoffmann-La Roche Ltd, Gilead Sciences Inc. and Merck KGaA are the major companies operating in the Hepatitis Therapeutics Market.
Which is the fastest growing region in Hepatitis Therapeutics Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Hepatitis Therapeutics Market?
In 2024, the North America accounts for the largest market share in Hepatitis Therapeutics Market.
What years does this Hepatitis Therapeutics Market cover?
The report covers the Hepatitis Therapeutics Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Hepatitis Therapeutics Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Hepatitis B Treatment Industry Report
Statistics for the 2024 Hepatitis B Treatment market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Hepatitis B Treatment analysis includes a market forecast outlook 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
