Neurology Devices Market Size
| Study Period | 2019 - 2029 |
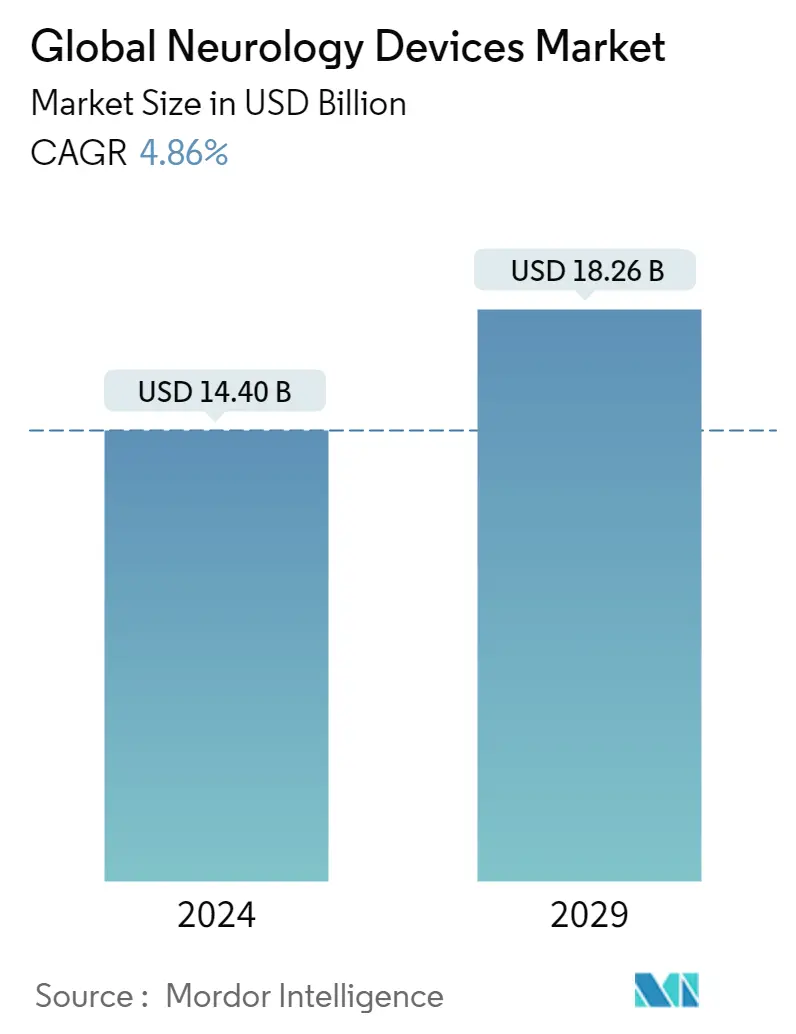
| Market Size (2024) | USD 14.40 Billion |
| Market Size (2029) | USD 18.26 Billion |
| CAGR (2024 - 2029) | 4.86 % |
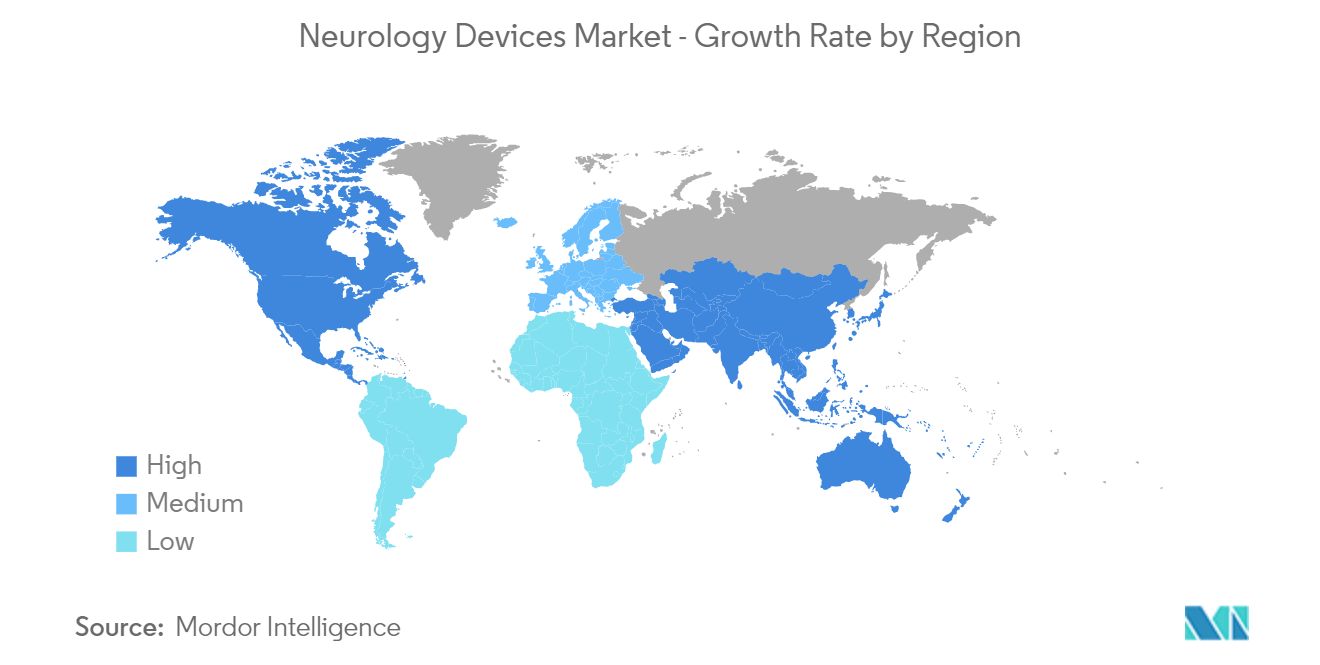
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Neurology Devices Market Analysis
The Global Neurology Devices Market size is estimated at USD 14.40 billion in 2024, and is expected to reach USD 18.26 billion by 2029, growing at a CAGR of 4.86% during the forecast period (2024-2029).
The COVID-19 pandemic had a significant impact on the global healthcare systems, with significant consequences not only for COVID-19-infected patients but also for others, resulting in the cancellation of treatment and surgical procedures to manage and reserve resources. For instance, in June 2020, the World Health Organization issued the results of a survey conducted by 155 countries for three weeks in May 2020, which showed that the prevention and treatment services of various chronic diseases, including various neurodegenerative disorders, such as Parkinson's, Alzheimer, and dementia, had been severely disrupted, and this has become a significant concern because people living with chronic diseases are at higher risk of severe COVID-19-related illness and death. In addition, 53% of the countries reported partial or complete disruption of services. This disruption in the management of different neurological conditions has impacted the lives of the patients. Moreover, the pandemic can also have a negative impact on new product launches, which are estimated to be the most prominent factor in the growth of the market studied. For instance, in April 2020, Biogen delayed Alzheimer's and dementia drug filing to Food and Drug Administration (FDA) while citing the complexity of data analysis and the impact of COVID-19 on its R&D resources. With the resumed services, treatment and surgical procedures and increased hospital visits are likely to increase the demand for neurology devices and treatment for various neurological disorders, thereby impacting the market growth.
The factors propelling the growth of the market are an increase in the incidence of neurological disorders, huge investments by private players in neurology devices, increase in research and development in the field of Neurotherapy.
The increasing burden of neurological disorders among the population is the key factor driving the demand for neurology devices over the forecast period. For instance, according to data published by the United Nations, in February 2021, about 1 billion people, nearly one in six of the world's population, suffer from neurological disorders, such as Alzheimer's and Parkinson's disease, strokes, multiple sclerosis, epilepsy migraine, brain injuries, and neuro infections in 2020. Additionally, as per the same source 62 million suffered from cerebrovascular disease, 50 million people suffered from epilepsy, 326 million from migraine, and 24 million from Alzheimer's disease and other dementias in 2019. Thus, the rising prevalence of neurological disorders among the population is expected to increase the demand for neurology devices to restore hearing and sight and provide an increased function for limb loss or congenital limb differences, thereby expected to augment the market growth.
Furthermore, the rising awareness among the population about neurological diseases and their treatment options is expected to drive market growth. For instance, as per a report published by World Health Organization (WHO), in April 2022, titled 'Draft Intersectoral Global Action Plan on Epilepsy and Other Neurological Disorders 2022-2031' 100% of countries are expected to have at least one functioning awareness campaign or advocacy program for neurological disorders by 2031. Such initiatives by the renowned organization are expected to drive the market in the coming future.
However, the high cost of equipment and stringent Food and Drug Administration validation and guidelines for new devices are likely to restrain the growth of the market.
Neurology Devices Market Trends
This section covers the major market trends shaping the Neurology Devices Market according to our research experts:
Neurostimulation Devices Segment Expects to Register a High CAGR in the Forecast Period
The neurostimulation devices segment is expected to witness significant growth over the forecast period owing to the factors such as the increasing prevalence of neurological disorders coupled with an increase in the aging population and lifestyle-related disorders such as depression and chronic pain and increasing adoption of technologically advanced products.
In addition, according to a study published by IEEE Engineering in Medicine and Biology Society, in February 2021, titled "New Advances in Neurostimulation for Chronic Pain", it has been found that Mainstay Medical designed a ReActiv8 implantable restorative neurostimulation system, which is used to treat intractable chronic lower back pain. Thus, the increasing advances in the products are expected to rise the adoption of these devices, thereby propelling the market growth.
Moreover, the increasing product launches and technological advancements in pain management are expected to increase the demand for neurostimulation devices, thereby fueling market growth. For instance, In July 2021, Medtronic launched the Percept PC Deep Brain Stimulation (DBS) system. The system integrated with BrainSense technology is made to administer therapeutic stimulation both inside and outside of the clinic while simultaneously capturing brain signals. The Percept PC device is indicated for individuals with Parkinson's disease, tremor, dystonia, and epilepsy and provides enhanced comfort.
Thus, rising product advancements and increasing product launches are expected to increase the demand for the studied segment, thereby boosting the market growth.
North America is Expected to Have the Significant Market Share
North America is expected to have a significant major market share in the neurology devices market owing to the factors such as the rising prevalence of neurological disorders, increasing funding from various organizations regarding the development for the treatment of neurodegenerative diseases, and rising research and development activities in the region.
The increasing burden of neurological disorders such as Parkinson's diseases, Alzheimer's diseases, epilepsy, neuromuscular disorders, and others is the key factor driving the growth of the market. For instance, according to data from Alzheimers.org, 6.2 million Americans aged 65 and older were thought to have Alzheimer's-related dementia in the United States in 2021. Also, the Parkinson's Foundation estimated that nearly 1 million Americans were living with Parkinson's disease (PD) in 2020. As per the same source, nearly 60,000 Americans are diagnosed with Parkinson's disease every year. Similarly, as per the Government of Canada Statistics report, as of July 2021, around 18.5% of the Canadian population was aged above 65 years which is an increase from 17.5% in July 2020. Thus, the rising burden of neurological diseases among the population is expected to increase the demand for early diagnosis and treatment, further increasing the demand for neurology devices, thereby propelling the market growth.
In addition, the growing geriatric population and the presence of robust healthcare infrastructure for neurological diagnostics and treatment are also contributing to the growth of the neurology devices market over the forecast period. For instance, according to the World Population Ageing 2021 report, the Mexican population aged 65 years and older is projected to reach 9.31 million by 2030. Thus, the increasing aging population is more prone to develop neurological disorders which are expected to increase the demand for various neurology devices for diagnosing and treatment of the conditions, which is expected to fuel the market growth.
Moreover, the rising company initiatives to develop neurology devices in the region are also contributing to the growth of the market. For instance, in January 2021, the United States Food and Drug Administration granted clearance to ClearMind Biomedical's neuro endoscope, the Axonpen System. This system can be used to illuminate and visualize intracranial tissue and fluids as well as the controlled tissue and/or fluid aspiration during surgery.
Thus, the increasing prevalence of neurological disorders, growing geriatric population, and increasing company activities in the region are expected to augment the market growth over the forecast period.
Neurology Devices Industry Overview
There are market participants involved in research and development for the development of advanced simulation and neuromodulation techniques with minimally invasive procedures that are preferable to the patients. Furthermore, many private companies are showing great interest in developing novel neurological devices to earn huge profits. Some of the key players in the neurology devices market are B. Braun Melsungen AG, Boston Scientific Corporation, Stryker Corporation, Medtronic PLC, Abbott Laboratories, and Smith & Nephew.
Neurology Devices Market Leaders
Boston Scientific Corporation
Stryker Corporation
B. Braun Melsungen AG
Medtronic PLC
Abbott Laboratories
*Disclaimer: Major Players sorted in no particular order
Neurology Devices Market News
In April 2022, Abbott launched an upgraded version of its digital health app, NeuroSphere myPath. The app's functionality has been enhanced so that it may better assist physicians in monitoring their patients as they utilize Abbott neurostimulation devices to treat their chronic pain.
In March 2021, Abbott announced the launch of the newly-approved NeuroSphere Virtual Clinic by which deep brain stimulation patients can have their devices programmed and reset remotely, from the comfort of their homes without having to travel to a specialist.
In January 2021, Medtronic announced that it made the first enrollment in the Adapt-PD trial for adaptive deep brain stimulation.
Neurology Devices Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increase in Incidence of Neurological Disorders
4.2.2 Huge Investments by Private Players in Neurology Devices
4.2.3 Increase in R&D in the Field of Neurotherapies
4.3 Market Restraints
4.3.1 High Cost of Equipment
4.3.2 Stringent FDA Validation and Guidelines for New Devices
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Type of Device
5.1.1 Cerebrospinal Fluid Management Devices
5.1.2 Interventional Neurology Devices
5.1.2.1 Interventional/Surgical Simulators
5.1.2.2 Neurothrombectomy Devices
5.1.2.3 Carotid Artery Stents
5.1.2.4 Embolic Coils
5.1.2.5 Support Devices
5.1.3 Neurosurgery Devices
5.1.3.1 Neuroendoscopes
5.1.3.2 Stereotactic Systems
5.1.3.3 Aneurysm Clips
5.1.3.4 Other Neurosurgery Devices
5.1.4 Neurostimulation Devices
5.1.4.1 Spinal Cord Stimulation Devices
5.1.4.2 Deep Brain Stimulation Devices
5.1.4.3 Sacral Nerve Stimulation Devices
5.1.4.4 Other Neurostimulation Devices
5.1.5 Other Types of Devices
5.2 Geography
5.2.1 North America
5.2.1.1 United States
5.2.1.2 Canada
5.2.1.3 Mexico
5.2.2 Europe
5.2.2.1 Germany
5.2.2.2 United Kingdom
5.2.2.3 France
5.2.2.4 Italy
5.2.2.5 Spain
5.2.2.6 Rest of Europe
5.2.3 Asia-Pacific
5.2.3.1 China
5.2.3.2 Japan
5.2.3.3 India
5.2.3.4 Australia
5.2.3.5 South korea
5.2.3.6 Rest of Asia-Pacific
5.2.4 Middle-East and Africa
5.2.4.1 GCC
5.2.4.2 South Africa
5.2.4.3 Rest of Middle-East and Africa
5.2.5 South America
5.2.5.1 Brazil
5.2.5.2 Argentina
5.2.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 B. Braun Melsungen AG
6.1.2 Boston Scientific Corporation
6.1.3 Stryker Corporation
6.1.4 Medtronic PLC
6.1.5 Abbott Laboratories
6.1.6 Johnson and Johnson
6.1.7 Smith & Nephew
6.1.8 MicroPort Scientific Corporation
6.1.9 Nihon Kohden Corporation
6.1.10 Penumbra Inc.
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Neurology Devices Industry Segmentation
As per the scope of the report, neurological devices are medical devices that help to diagnose, prevent, and treat a variety of neurological disorders and conditions, such as Alzheimer's disease, Parkinson's disease, major depression, and traumatic brain injury. The Neurology Devices Market is segmented by Type of Device (Cerebrospinal Fluid Management Devices, Interventional Neurology Devices, Neurosurgery Devices, Neurostimulation Devices, and Other Types of Devices) and Geography (North America, Europe, Asia-Pacific, Middle East, and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions, globally. The report offers the value (in USD million) for the above segments.
| By Type of Device | |||||||
| Cerebrospinal Fluid Management Devices | |||||||
| |||||||
| |||||||
| |||||||
| Other Types of Devices |
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Neurology Devices Market Research FAQs
How big is the Global Neurology Devices Market?
The Global Neurology Devices Market size is expected to reach USD 14.40 billion in 2024 and grow at a CAGR of 4.86% to reach USD 18.26 billion by 2029.
What is the current Global Neurology Devices Market size?
In 2024, the Global Neurology Devices Market size is expected to reach USD 14.40 billion.
Who are the key players in Global Neurology Devices Market?
Boston Scientific Corporation, Stryker Corporation, B. Braun Melsungen AG, Medtronic PLC and Abbott Laboratories are the major companies operating in the Global Neurology Devices Market.
Which is the fastest growing region in Global Neurology Devices Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Global Neurology Devices Market?
In 2024, the North America accounts for the largest market share in Global Neurology Devices Market.
What years does this Global Neurology Devices Market cover, and what was the market size in 2023?
In 2023, the Global Neurology Devices Market size was estimated at USD 13.73 billion. The report covers the Global Neurology Devices Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Global Neurology Devices Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Neurology Device Industry Report
Statistics for the 2024 Neurology Device market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Neurology Device analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
