Traveler's Diarrhea Treatment Market Size
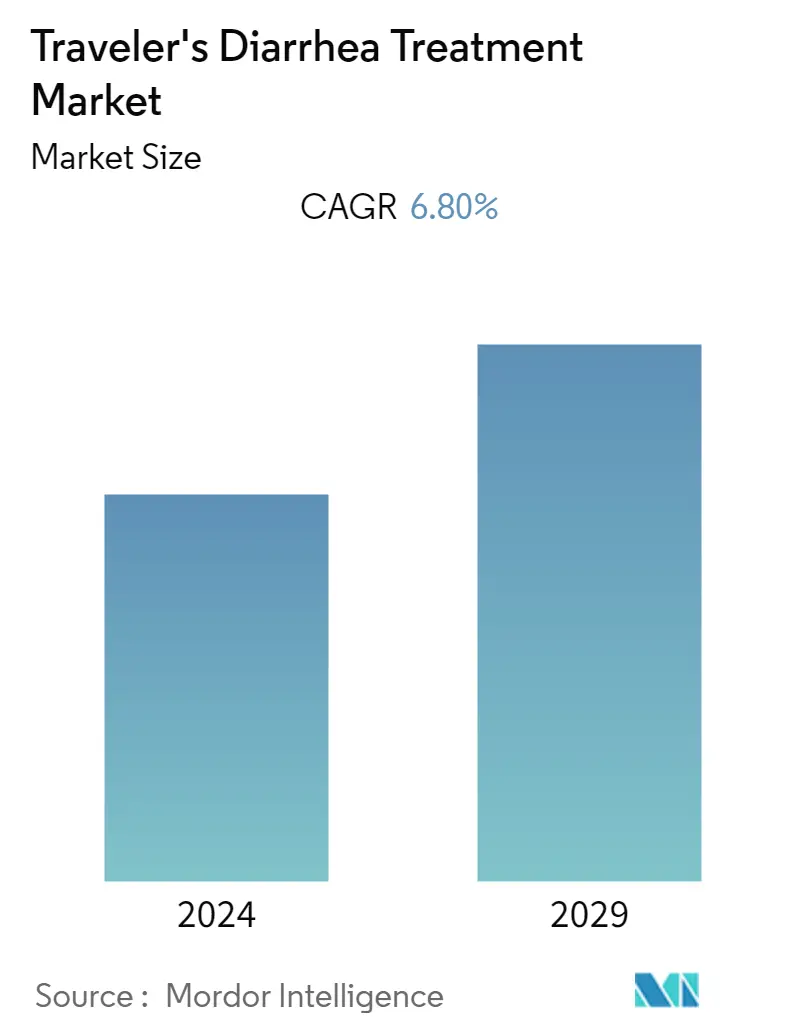
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
| CAGR | 6.80 % |
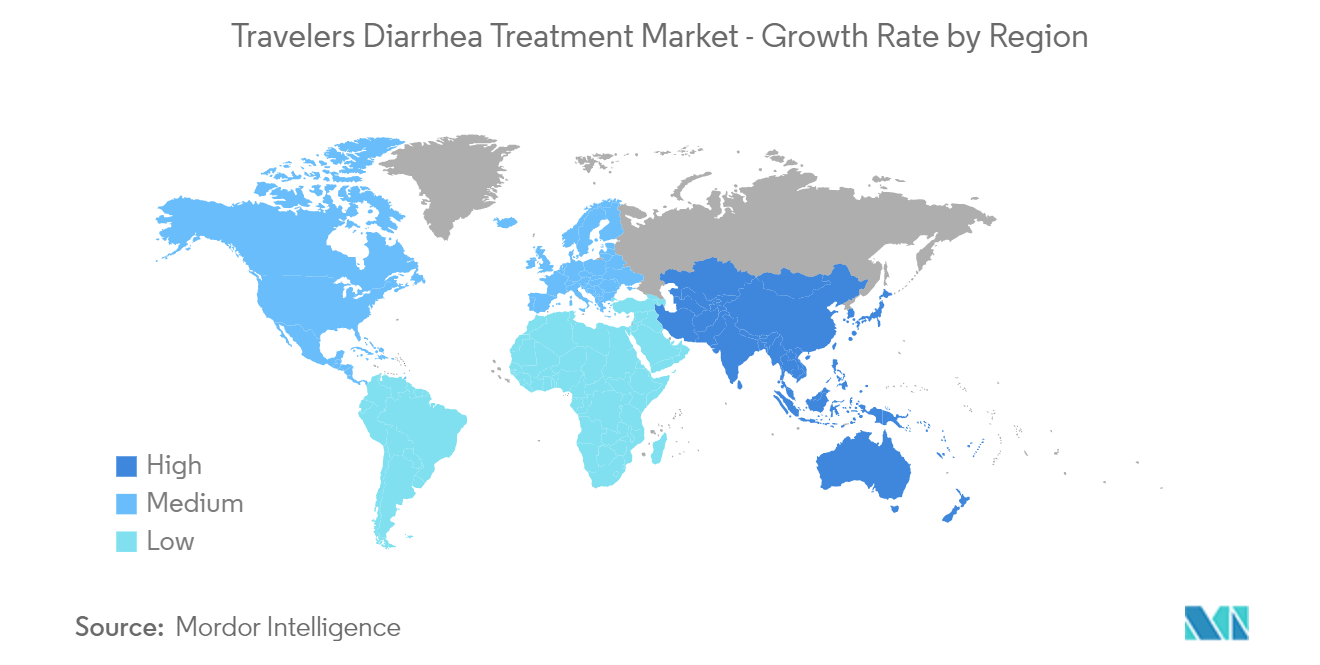
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Traveler's Diarrhea Treatment Market Analysis
The traveler's diarrhea treatment market is expected to register a CAGR of 6.8% during the forecast period.
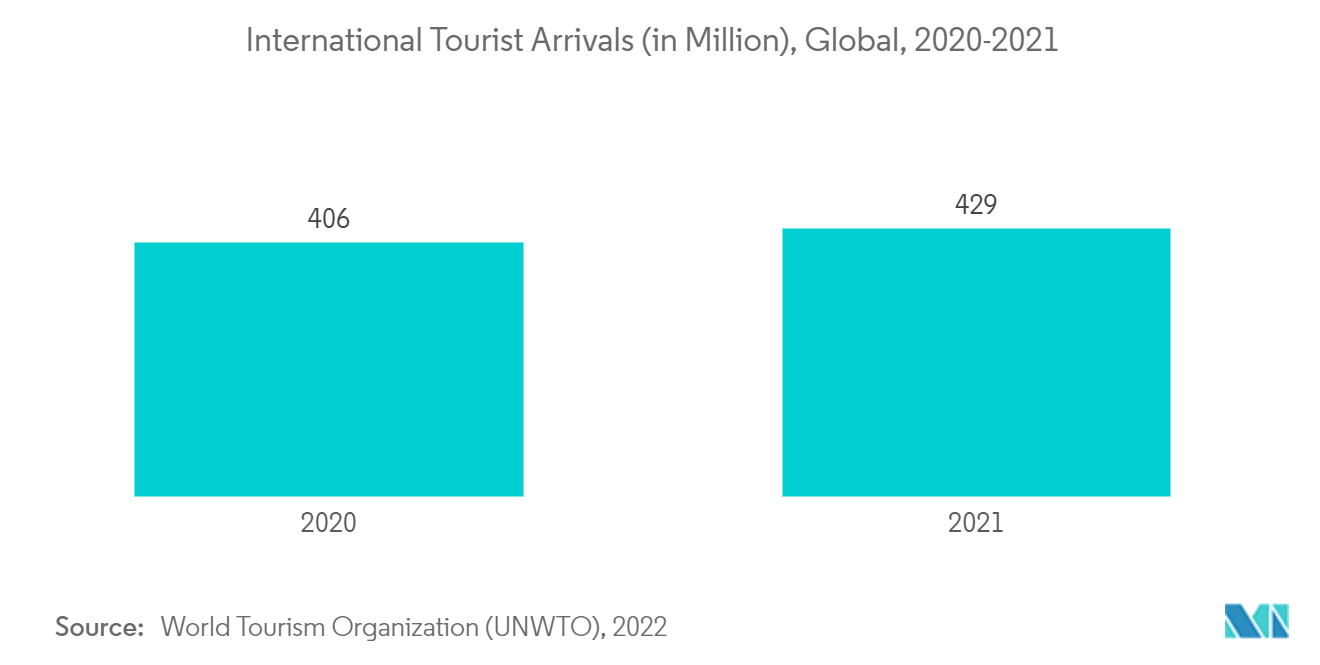
The pandemic caused unprecedented disruption in the tourism industry, with a massive fall in international demand amid widespread lockdowns and travel restrictions by countries to contain the spread of the virus, which impacted the traveler's diarrhea market. For instance, according to the World Tourism Organization (UNWTO), in 2022, the pandemic caused a 72% decline in international tourist arrivals in 2020 and 71% in 2021. Thus, a decrease in tourism significantly impacted the market's growth. However, the market is currently growing at a stable pace due to the resumption of tourism. According to the World Tourism Organization, tourism doubled in 2022. It is expected to witness a similar trend over the coming years. Thus, the market is expected to gain traction over the coming years because of declining COVID-19 cases and increased tourism.
Traveler's diarrhea affects millions of international travelers traveling to developing countries yearly. Food and contaminated water are the primary sources of infection. Bacteria, such as E. coli and Salmonella, are the common causes of traveler's diarrhea. According to the UNWTO, between January and September 2022, a projected 700 million tourists traveled abroad, more than double (+133%) the number seen during the same period in 2021. In addition, Europe took the lead in the recovery of global travel, with numbers 81% higher than before the pandemic. Between January and September 2022, international arrivals to the Middle East increased by 225%. The prevalence of traveler's diarrhea is higher among travelers visiting regions in South Asia and Southeast Asia. The travel destination is the most significant risk factor for traveler's diarrhea.
According to the article published in the June 2021 issue of the National Library of Medicine, traveler's diarrhea is a common ailment in individuals traveling to resource-limited destinations overseas. It is estimated to affect nearly 40-60% of travelers and is the most common travel-associated condition. Thus, the significant burden of traveler's diarrhea is expected to boost the demand for its treatment, thereby boosting the market's growth.
Hence, owing to these factors, the market is likely to witness significant growth during the forecast period. However, the side effects associated with the drugs are expected to impede the market's growth.
Traveler's Diarrhea Treatment Market Trends
Antibiotics Segment is Expected to Hold Significant Share Over the Forecast Period
A course of antibiotics helps treat diarrhea caused by bacteria or parasites. Most of the time, over-the-counter drugs like antibiotics can help reduce the discomfort associated with traveler's diarrhea. For the treatment of severe traveler's diarrhea, azithromycin is preferred. Traveler's diarrhea that is severe and not dysentery-related may be treated with fluoroquinolones. Traveler's severe diarrhea and non-dysentery can be treated with rifaximin. Traveler's diarrhea can be treated with single-dose antibiotic regimens.
The increasing prevalence of traveler's diarrhea is expected to boost the segment's growth. For instance, according to the article published in Travel Medicine and Infectious Diseases in February 2022, travel-related diarrhea (TD) is the most common sickness among travelers from high-income countries to middle- or low-income countries. Every year, almost 80 million people who travel internationally get diarrhea. Because of this, TD has always been and still is a major issue on a global scale. Thus, the significant burden of traveler's diarrhea is expected to boost the demand for antibiotic drugs, thereby boosting the segment's growth.
According to the study published in Tropical Diseases and Infectious Diseases in November 2022, almost all medical experts participating in pre-travel counseling frequently advise clients to purchase antibiotics as preventative medicine. Rifaximin was accurately identified by about 60% of the participating physicians as a treatment option for severe, non-dysenteric TD and as a preventative option for patients at high risk for consequences. Thus, the significance of antibiotics in traveler's diarrhea treatment is expected to boost the demand for antibiotics, thereby boosting the market segment's growth.
North America Holds Significant Share in the Market and is Expected to Grow During the Forecast Period
The North American region holds the largest share of the market due to the increasing burden of traveler's diarrhea and the strong presence of manufacturers in this region. The rising awareness, along with the easy availability of over-the-counter medications, is likely to favor the growth of the market in North America.
In the United States, the growing number of international travelers visiting the country for leisure trips and the rising business travel within the country are driving the traveler's diarrhea treatment market. For instance, according to the US Department of Commerce report in June 2022, international visitors within the country increased from 19.2 million in 2020 to 22.1 million in 2021. Thus, increasing tourism in the United States may lead to more cases of traveler's diarrhea, which would ultimately boost the market's growth over the forecast period.
The strategic activities by the market players, such as product launches, mergers and acquisitions, and partnerships, are expected to propel the market's growth in the region over the forecast period. For instance, in December 2022, Immuron Limited announced that it had received approval from the US FDA to proceed with the clinical evaluation of Travelan. The Investigational New Drug (IND) application to evaluate the efficacy of a single dose of Travelan to prevent infectious diarrhea caused by Enterotoxigenic E.coli (ETEC) is now active.
Thus, all such factors are expected to boost the market in the region over the forecast period.
Traveler's Diarrhea Treatment Industry Overview
The traveler's diarrhea treatment market is moderately competitive, with the presence of several market players. The major players are Bausch Health Companies Inc. (Salix Pharmaceuticals), Bristol-Myers Squibb Company, Cosmo Pharmaceuticals NV, Immuron Limited, Johnson & Johnson, Pfizer Inc., Procter & Gamble, Sanofi SA, and Valneva Canada Inc.
Traveler's Diarrhea Treatment Market Leaders
Bausch Health Companies Inc. (Salix Pharmaceuticals)
Cosmo Pharmaceuticals N.V.
Johnson & Johnson
Pfizer Inc.
Procter & Gamble
*Disclaimer: Major Players sorted in no particular order
Traveler's Diarrhea Treatment Market News
- In September 2022, Bausch Health Companies Inc. and its gastroenterology business, Salix Pharmaceuticals, responded to the US FDA's tentative approval of Norwich Pharmaceuticals' rifaximin 200 mg product, XIFAXAN (rifaximin), indicated for travelers' diarrhea.
- In January 2022, Immuron Limited announced that it had been granted a European patent for compositions and methods for treating traveler's diarrhea.
Travelers Diarrhea Treatment Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Surge in the Number of Travelers
4.2.2 Rising Awareness Among Travelers About Hygiene, Food, and Water Safety
4.2.3 Availability of Over-the-counter Medications
4.3 Market Restraints
4.3.1 Side Effects Associated with the Drugs
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Drug Type
5.1.1 Antibiotics
5.1.2 Anti-diarrheal
5.1.3 Other Drug Types
5.2 By Distribution Channel
5.2.1 Hospital Pharmacies
5.2.2 Retail Pharmacies
5.2.3 Other Distribution Channels
5.3 By Geography
5.3.1 North America
5.3.1.1 United States
5.3.1.2 Canada
5.3.1.3 Mexico
5.3.2 Europe
5.3.2.1 Germany
5.3.2.2 United Kingdom
5.3.2.3 France
5.3.2.4 Italy
5.3.2.5 Spain
5.3.2.6 Rest of Europe
5.3.3 Asia-Pacific
5.3.3.1 China
5.3.3.2 Japan
5.3.3.3 India
5.3.3.4 Australia
5.3.3.5 South Korea
5.3.3.6 Rest of Asia-Pacific
5.3.4 Middle East and Africa
5.3.4.1 GCC
5.3.4.2 South Africa
5.3.4.3 Rest of Middle East and Africa
5.3.5 South America
5.3.5.1 Brazil
5.3.5.2 Argentina
5.3.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Bausch Health Companies Inc. (Salix Pharmaceuticals)
6.1.2 Bristol-Myers Squibb Company
6.1.3 Cosmo Pharmaceuticals NV
6.1.4 Immuron Limited
6.1.5 Johnson & Johnson
6.1.6 Pfizer Inc.
6.1.7 Procter & Gamble
6.1.8 Sanofi SA
6.1.9 Valneva Canada Inc.
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Traveler's Diarrhea Treatment Industry Segmentation
As per the scope of the report, traveler's diarrhea is a digestive tract disorder characterized by an intestinal infection that occurs when a person consumes contaminated food and water.
The traveler's diarrhea treatment market is segmented by drug type (antibiotics, anti-diarrheal, and other drug types), distribution channel (hotel pharmacies, retail pharmacies, and other distribution channels), and geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The report offers the value (in USD million) for the above segments. The market report also covers the estimated market sizes and trends for 17 countries across major global regions. The report offers the value (USD million) for the above segments.
| By Drug Type | |
| Antibiotics | |
| Anti-diarrheal | |
| Other Drug Types |
| By Distribution Channel | |
| Hospital Pharmacies | |
| Retail Pharmacies | |
| Other Distribution Channels |
| By Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Travelers Diarrhea Treatment Market Research FAQs
What is the current Traveler's Diarrhea Treatment Market size?
The Traveler's Diarrhea Treatment Market is projected to register a CAGR of 6.80% during the forecast period (2024-2029)
Who are the key players in Traveler's Diarrhea Treatment Market?
Bausch Health Companies Inc. (Salix Pharmaceuticals), Cosmo Pharmaceuticals N.V., Johnson & Johnson, Pfizer Inc. and Procter & Gamble are the major companies operating in the Traveler's Diarrhea Treatment Market.
Which is the fastest growing region in Traveler's Diarrhea Treatment Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Traveler's Diarrhea Treatment Market?
In 2024, the North America accounts for the largest market share in Traveler's Diarrhea Treatment Market.
What years does this Traveler's Diarrhea Treatment Market cover?
The report covers the Traveler's Diarrhea Treatment Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Traveler's Diarrhea Treatment Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Traveler's Diarrhea Treatment Industry Report
Statistics for the 2024 Traveler's Diarrhea Treatment market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Traveler's Diarrhea Treatment analysis includes a market forecast outlook to for 2024 to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
