Cryotherapy Market Size
| Study Period | 2019 - 2029 |
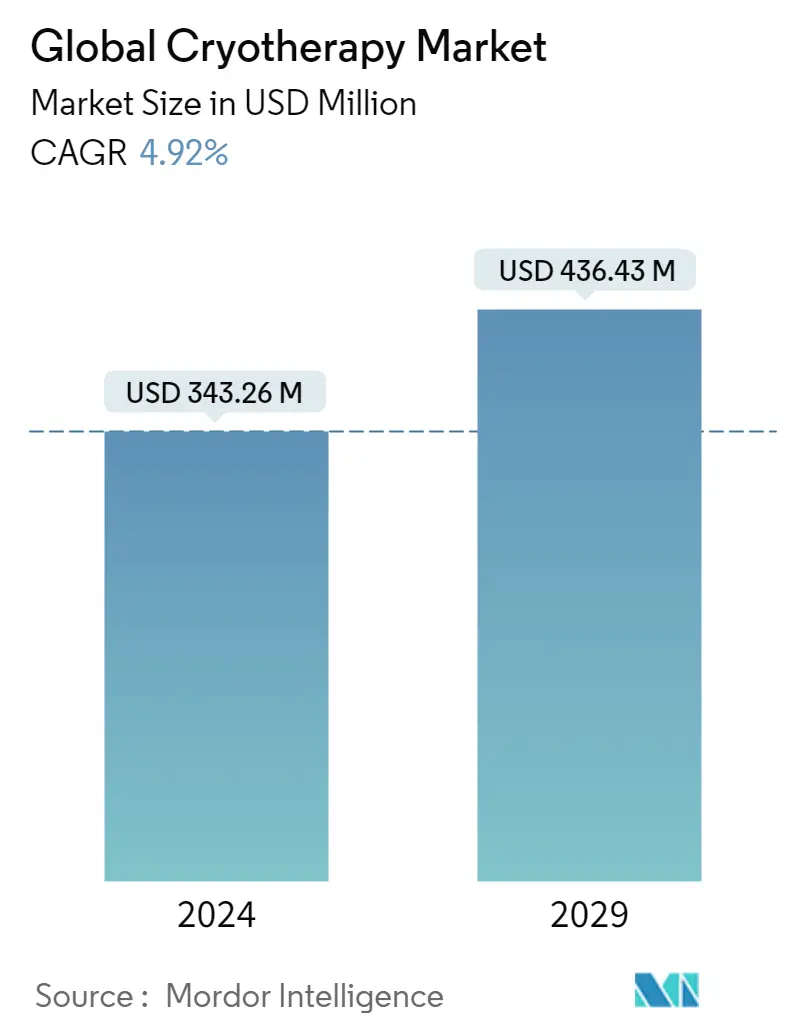
| Market Size (2024) | USD 343.26 Million |
| Market Size (2029) | USD 436.43 Million |
| CAGR (2024 - 2029) | 4.92 % |
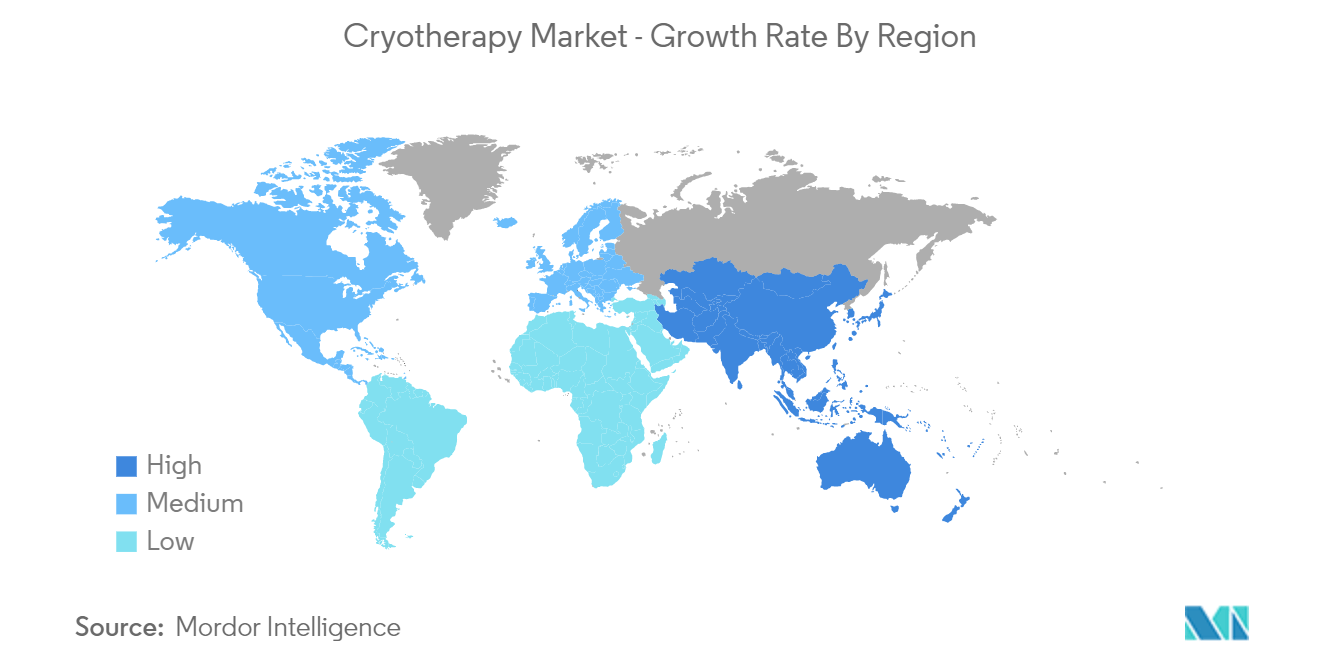
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Cryotherapy Market Analysis
The Global Cryotherapy Market size is estimated at USD 343.26 million in 2024, and is expected to reach USD 436.43 million by 2029, growing at a CAGR of 4.92% during the forecast period (2024-2029).
The outbreak of the COVID-19 pandemic impacted the cryotherapy market significantly. The surge in the number of cancer patients and other chronic disease patients amid pandemics has increased, which is attributed to a growth factor for the cryotherapy market. In addition, increased research for finding benefits of cryotherapy for Covid-19 treatment increased. For instance, the article 'Cryotherapy as a Treatment for Loss of Smell from Covid' was published in March 2022, where Bastien, spokesperson of the France-based cryotherapy center company Cryotera, mentioned that cryotherapy is increasingly being investigated due to its health benefits and researchers at the company suggested that the extreme cold treatment could help restore a sense of smell after Covid-19. Therefore, the pandemic is predicted to have a positive impact on the cryotherapy market growth.
Factors such as increasing applications of cryosurgery, technological advancements in cryotherapy equipment, and rising preference for minimally invasive techniques are major driving factors for the market studied.
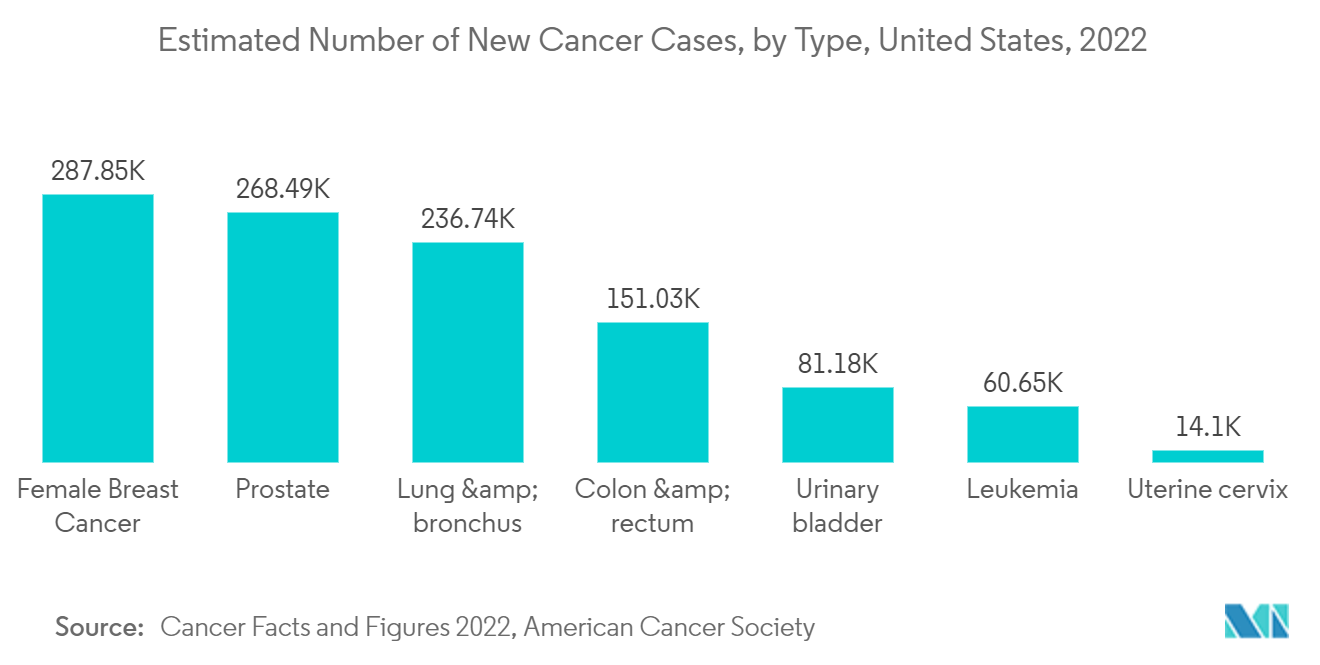
Furthermore, the growing number of cancer cases globally is primarily driving the studied market growth. The updated in February 2022 by the American Society of Clinical Oncology (ASCO), an estimated 324,635 people were diagnosed with melanoma 2020 worldwide, and in 2020, about 2,400 cases of melanoma were estimated to be diagnosed in people aged 15 to 29. Cryotherapy is often used for pre-cancerous conditions such as actinic keratosis and small basal cell and squamous cell carcinomas. Thus, the prevalence of skin cancer among the target population globally is augmenting the growth of the market studied.
Moreover, the increased advancements in the cryotherapy segment are predicted to drive the demand for skin cancer therapeutics over the forecast period. For instance, in February 2021, Hyderabad-based (India) SLG Hospitals launched Cryotherapy (cold therapy) and advanced physiotherapy services aimed at providing specialized treatment for sports injuries and post-operation pain management. Again, in December 2021, Medilink received Food and Drug Administration (FDA) approval for Portable cryotherapy Hydrozid and launched the Hybrizoid in the United States. In addition, in May 2022, iCRYO, one of the fast-growing recovery and wellness brands, announced its plan to open two new regional offices in Loveland, Colorado, and Dallas, Texas, to further support the rapid expansion and development of cryotherapy & recovery services. Thus, these increasing advancements are expected to bolster the growth of the studied market.
Therefore, the factors mentioned above are attributed collectively to the studied market growth over the forecast period. However, the hazardous effects of cryogenic gases and the complexity of the cryotherapy mechanisms are expected to hinder the market growth over the forecast period.
Cryotherapy Market Trends
This section covers the major market trends shaping the Cryotherapy Market according to our research experts:
Oncology by Segment is Expected to Grow Over the Forecast Period
Cryotherapy is used to treat several types of cancers, as well as some precancerous or noncancerous conditions. In addition to prostate and liver tumors, cryosurgery can be an effective treatment for Retinoblastoma, early-stage skin cancers (both basal cell and squamous cell carcinomas), precancerous skin growths known as actinic keratosis, and precancerous conditions of the cervix known as cervical intraepithelial neoplasia. The oncology segment may hold the highest market share during the forecast period due to the high incidence and prevalence of cancer, as well as growing awareness regarding various types of cryotherapy approaches.
Minimally invasive ablative procedures have emerged as popular alternatives for cancer treatment or surgical tumor removal with rapid advances in modern imaging. Tumor ablation modalities are currently offered in clinical practice, and cryoablation procedures are extensively used. Increasing research for analyzing the impact of cryotherapy on cancer treatment is also increasing. For instance, in February 2022 research article titled 'Recent progress in cryoablation cancer therapy and nanoparticles mediated cryoablation' was published in Theranostics journal, which was focused on recent progress in cryoablation cancer therapy and nanoparticles mediated cryoablation. As per the research, image-guided combination cryotherapy has the possibility of eradicating cancer while preserving neighboring organs and tissues: also, the research paper mentioned above stated that recent development of multifunctional nanocarriers can play a major role in the expansion of cryoablation in the treatment of cancer. Thus, the extensive and rising research on the benefits of cryotherapy and combination cryoablation cancer therapies is predicted to create a better opportunity for the oncology segment.
The insights on increased R&D in the segment are also predicted to drive the segment growth. For instance, in April 2021, IceCure Medical Ltd., developer of the next generation cryoablation technology, received Breakthrough Device Designation from Food and Drug Administration (FDA) for its lead asset, ProSense, and proposed indication for use, including for use in the treatment of patients with T1 invasive breast cancer and/or patients not suitable for surgical alternatives for the treatment of breast cancer. ProSense is a liquid-nitrogen-based cryoablation system that enables minimally-invasive treatment of cancer tumors.
Therefore, owing to the factors stated above, the oncology segment is growing, and it is expected to have significant growth over the forecast period. Hence, driving the growth of the studied market.
North America is Expected to Dominate the Market Over the Forecast Period
North America is the largest regional market in terms of revenue, owing to high healthcare expenditure by local governments, highly developed healthcare infrastructure in the United States (US) and Canada, and the availability of well-developed cryotherapy products in this region may also influence the market growth during the forecast period.
Along with the healthcare expenditure, the presence of skilled professionals, increasing adoption of whole-body cryotherapy, increasing adoption of cryotherapy for cancers and skin disorders, rising prevalence of cancer, and increasing adoption of minimally invasive surgeries are primary and major driving factors for the overall market growth in the country. For example, the Cancer Facts & Figures 2022 published by the American Cancer Society shows that there will be an estimated 1.9 million new cancer cases to be diagnosed in the US. And as per the source mentioned above, Cancer is the second most common cause of death in the US, after heart disease. Due to the benefits of cryotherapy in cancer treatment and tumor removal by surgery, the cryotherapy market is predicted to grow in the country.
Moreover, the intended involvement of market players to develop and create demand for cryotherapy in the country is predicted to drive market growth. For instance, in December 2021, Restore Hyper Wellness (Restore), one of the leading providers of proactive wellness solutions, invested USD 140 million, led by General Atlantic, a leading global growth equity firm. Restore's most popular modalities include IV Drip Therapy and Whole-Body Cryotherapy. Further, in July 2020, Channel Medsystems, Inc., a medical device company, launched its Cerene Cryotherapy treatment for heavy menstrual bleeding. Hence, the recent advancement in the United States is garnering market growth in the country.
Hence, as per the factors mentioned above, the North American region is anticipated to create an opportunity for cryotherapy, driving the overall market growth in the country.
Cryotherapy Industry Overview
The cryotherapy market is competitive, and the market consists of several major players who are engaged in continuous product development and launches. Some companies currently dominating the market are Medtronic, Impact Cryotherapy, Zimmer MedizinSysteme GmbH, Metrum Cryoflex Polska Sp. z o.o., and Brymill Cryogenic Systems, among others.
Cryotherapy Market Leaders
Impact Cryotherapy
Quantum Cryogenics
CryoConcepts LP.
Zimmer MedizinSysteme GmbH
Boston Scientific Corporation
*Disclaimer: Major Players sorted in no particular order
Cryotherapy Market News
- In May 2022, The Bulgari SPA London, a wellness center, presented four revolutionary cryotherapy treatments suitable for both men and women. The Cryotherapy Sports Massage targets specific areas to alleviate pain, inflammation and speed up recovery from injuries.
- In April 2022, IceCure Medical Ltd., developer of minimally-invasive cryoablation technology, the ProSense System, that destroys tumors by freezing as an alternative to surgical tumor removal, hosted three key ProSense System events at the European Conference on Interventional Oncology (ECIO) in Vienna, Austria.
Cryotherapy Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increasing Applications of Cryosurgery
4.2.2 Technological Advancements in Cryotherapy Equipment
4.2.3 Rising Preference for Minimally Invasive Techniques
4.3 Market Restraints
4.3.1 Hazardous Effects of Cryogenic Gases
4.3.2 Complexity of the Cryotherapy Mechanisms
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Product
5.1.1 Cryosurgery Devices
5.1.1.1 Tissue Contact Probes
5.1.1.2 Tissue Spray Probes
5.1.1.3 Epidermal and Subcutaneous Cryoablation Devices
5.1.2 Localized Cryotherapy Devices
5.1.3 Cryochambers and Cryosaunas
5.2 By Application
5.2.1 Surgical Applications
5.2.1.1 Oncology
5.2.1.2 Cardiology
5.2.1.3 Dermatology
5.2.1.4 Other Surgical Applications
5.2.2 Pain Management
5.2.3 Recovery, Health, and Beauty
5.3 By End User
5.3.1 Hospitals
5.3.2 Cryotherapy Centers
5.4 Geography
5.4.1 North America
5.4.1.1 United States
5.4.1.2 Canada
5.4.1.3 Mexico
5.4.2 Europe
5.4.2.1 Germany
5.4.2.2 United Kingdom
5.4.2.3 France
5.4.2.4 Italy
5.4.2.5 Spain
5.4.2.6 Rest of Europe
5.4.3 Asia-Pacific
5.4.3.1 China
5.4.3.2 Japan
5.4.3.3 India
5.4.3.4 Australia
5.4.3.5 South Korea
5.4.3.6 Rest of Asia-Pacific
5.4.4 Middle-East and Africa
5.4.4.1 GCC
5.4.4.2 South Africa
5.4.4.3 Rest of Middle-East and Africa
5.4.5 South America
5.4.5.1 Brazil
5.4.5.2 Argentina
5.4.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Medtronic
6.1.2 Impact Cryotherapy
6.1.3 Zimmer MedizinSysteme GmbH
6.1.4 METRUM CRYOFLEX POLSKA Sp. z o.o.
6.1.5 Brymill Cryogenic Systems
6.1.6 CryoConcepts LP.
6.1.7 Kriosystem Life
6.1.8 Boston Scientific Corporation
6.1.9 CooperSurgical Inc.
6.1.10 Quantum Cryogenics
6.1.11 IceCure Medical
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Cryotherapy Industry Segmentation
As per the scope of the report, cryotherapy is also known as cryoablation, cryosurgery, targeted cryoablation therapy, and percutaneous cryotherapy. Cryotherapy is a minimally invasive method that uses extreme cold to freeze and destroy the infected tissues or cells. The cryotherapy market is segmented by Product (Cryosurgery Devices (Tissue Contact Probes, Tissue Spray Probes, and Epidermal and Subcutaneous Cryoablation Devices), Localized Cryotherapy Devices, Cryochambers, and Cryosaunas), Application (Surgical Applications (Oncology, Cardiology, Dermatology and Other Surgical Applications), Pain Management and Recovery, Health, and Beauty) End User (Hospitals and Cryotherapy Centers), and Geography (North America, Europe, Asia-Pacific, Middle-East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions, globally. The report offers the value (USD million) for the above segments.
| By Product | |||||
| |||||
| Localized Cryotherapy Devices | |||||
| Cryochambers and Cryosaunas |
| By Application | ||||||
| ||||||
| Pain Management | ||||||
| Recovery, Health, and Beauty |
| By End User | |
| Hospitals | |
| Cryotherapy Centers |
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Cryotherapy Market Research FAQs
How big is the Global Cryotherapy Market?
The Global Cryotherapy Market size is expected to reach USD 343.26 million in 2024 and grow at a CAGR of 4.92% to reach USD 436.43 million by 2029.
What is the current Global Cryotherapy Market size?
In 2024, the Global Cryotherapy Market size is expected to reach USD 343.26 million.
Who are the key players in Global Cryotherapy Market?
Impact Cryotherapy, Quantum Cryogenics , CryoConcepts LP., Zimmer MedizinSysteme GmbH and Boston Scientific Corporation are the major companies operating in the Global Cryotherapy Market.
Which is the fastest growing region in Global Cryotherapy Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Global Cryotherapy Market?
In 2024, the North America accounts for the largest market share in Global Cryotherapy Market.
What years does this Global Cryotherapy Market cover, and what was the market size in 2023?
In 2023, the Global Cryotherapy Market size was estimated at USD 327.16 million. The report covers the Global Cryotherapy Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Global Cryotherapy Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Cryotherapy Industry Report
Statistics for the 2024 Cryotherapy market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Cryotherapy analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
