Cardiovascular Devices Market Size
| Study Period | 2021 - 2029 |
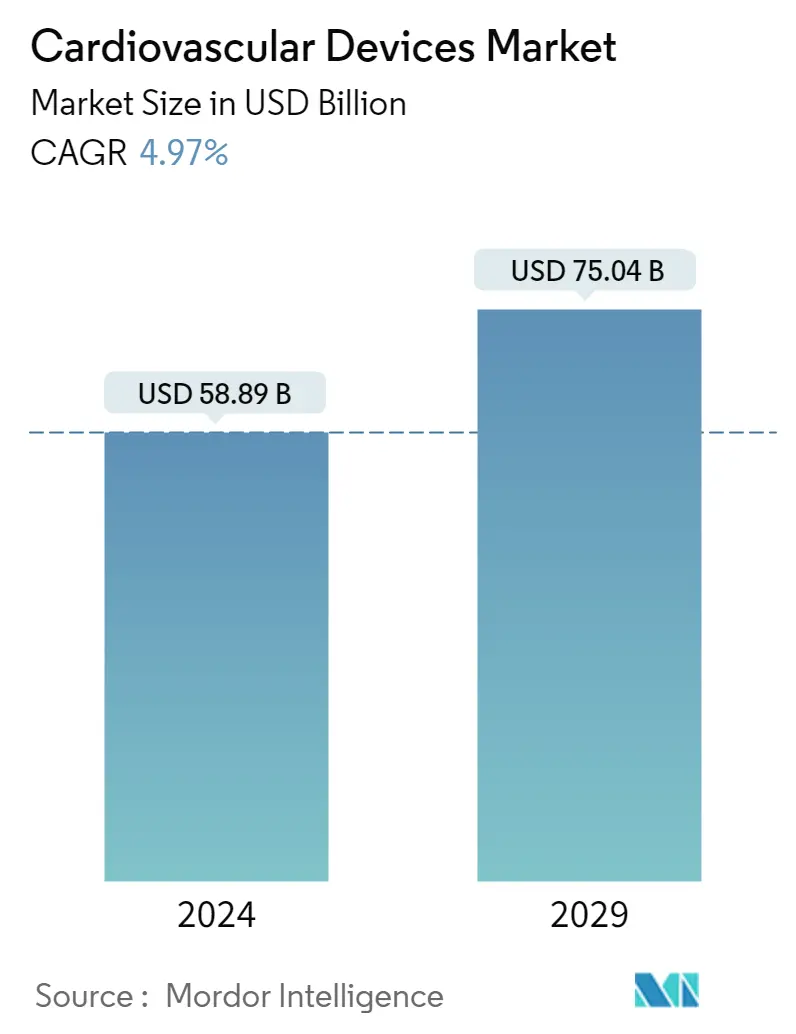
| Market Size (2024) | USD 58.89 Billion |
| Market Size (2029) | USD 75.04 Billion |
| CAGR (2024 - 2029) | 4.97 % |
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Cardiovascular Devices Market Analysis
The Cardiovascular Devices Market size is estimated at USD 58.89 billion in 2024, and is expected to reach USD 75.04 billion by 2029, growing at a CAGR of 4.97% during the forecast period (2024-2029).
The emergence of the COVID-19 pandemic hurt the cardiovascular devices market in the beginning. It had a detrimental effect on the market due to the reduction in the diagnosis of cardiovascular diseases due to healthcare resources being reserved for COVID-19 patients. Many medical device companies have experienced losses in their cardiovascular device businesses due to the pandemic. However, production and healthcare facilities got resumed which is expected to have a positive impact on the market. For instance, as per the 2022 annual report of Medtronic, a technology company having a major share of the revenue from the cardiovascular portfolio has increased revenue due to the global procedure volumes recovering from the decline brought on by the COVID-19 pandemic in the first and second quarters of the fiscal year 2021.
The major factors driving the market are rapid technological advancements, the rising global burden of various cardiovascular diseases, and increased preference for minimally invasive procedures. Heart-related mortality is caused by the rising prevalence of heart disorders such as cardiomyopathy and stroke. The most common diseases in the world are those related to the cardiovascular system. For instance, per the report published by the Heart and Stroke Foundation of Canada in February 2022, there are 750,000 patients diagnosed with heart failure and this is increasing every year. Hence for proper diagnosis and treatment of heart-related diseases cardiovascular devices play an important role that drives the market growth over the study period.
Moreover, technologies used in the development of cardiology devices have improved significantly over the past decade, and indications for these devices have expanded. This has led to an increasing number of patients being managed with cardiology devices, resulting in exponential therapeutical and monitoring outcomes. Artificial intelligence is one such advancement that has a significant positive impact on cardiology with improved capabilities for monitoring certain heart conditions. For instance, in July 2021, Medtronic received United States FDA clearance for two AccuRhythm AI algorithms for use with the LINQ II insertable cardiac monitor (ICM). AccuRhythm AI applies artificial intelligence (AI) to heart rhythm event data collected by LINQ II, improving the accuracy of information physicians receive so they can better diagnose and treat abnormal heart rhythms.
However, the stringent regulatory policies and high cost of instruments and procedures are the factors restraining the growth of the cardiovascular devices market.
Cardiovascular Devices Market Trends
This section covers the major market trends shaping the Cardiovascular Devices Market according to our research experts:
The Electrocardiogram (ECG) Under Diagnostic and Monitoring Devices Segment is Expected to Hold a Significant Share in the Market
More people are using ECGs for diagnostic and monitoring purposes thanks to the latest generation of lightweight and compact devices, which are especially popular in the home healthcare market. Rising rates of cardiovascular disease (CVD) necessitate continuous ECG monitoring. In addition, the development of wireless ECG has allowed for real-time patient monitoring and diagnosis, which has contributed to the growth of the market. In January 2022, Philips, for instance, began offering a 12-lead electrocardiogram (ECG) solution for use in decentralized clinical trials that could be performed in the comfort of a patient's own home.
Among the new technologies in cardiac devices, wearable smart devices equipped with heart-monitoring capabilities, such as bands, watches, and rings, have become the norm for many people globally. In February 2021, VivaLNK launched a wearable ECG monitoring solution designed to wirelessly capture ECG and heart rate during a six-minute walk test (6MWT). Thus the new product launches and approvals increase the scope for the usage of ECG which drives the market segment over the study period.
Additionally, the collaborations among the major players in the cardiovascular market help the segment to grow over the study period. For instance, in October 2022, AccurKardia is going to collaborate with Mawi, a medtech provider of medical-grade wearables in the cardiac space, to integrate its proprietary ECG analytics into Mawi's new cardiac monitoring watch. This collaboration highlights AccurKardia's commitment to enabling world-class cardiac care through remote patient monitoring solutions.
Therefore, due to the aforementioned factors, this market segment is expected to witness significant growth during the forecast period.
North America is Expected to Hold a Significant Share in the Market During the Forecast Period
North America is expected to dominate the cardiovascular devices market due to the high prevalence of cardiovascular diseases, the high adoption rate of minimally invasive procedures, the presence of reimbursements, the rising geriatric population, and the high demand for continuous and home-based monitoring.
According to the CDC updated report published in October 2022, for every 40 seconds, a heart attack occurs in the United States, and also about 805,000 Americans experience a heart attack each year. Hence the rising burden of cardiovascular diseases in coming years increases the demand for cardiovascular diagnostics and treatment devices. Additionally, a few of the key market players in the country are developing novel products and technologies to compete with the existing products, while others are acquiring and partnering with other companies trending in the market.
For instance, in April 2022, Abbott received approval from the United States FDA for its Aveir single-chamber (VR) leadless pacemaker for the treatment of patients in the United States with slow heart rhythms. This marks a significant advancement in patient care and brings new, never-before-seen features to patients and their physicians. Similarly, in February 2022, Abbott received expanded indication approval from the United States FDA for the CardioMEMS HF System to support the care of patients suffering from earlier stages of heart failure. These new product approvals increase the scope for the usage of devices that drive the market in the region.
Furthermore, supportive awareness initiatives to create disease awareness are projected to support the market for cardiovascular devices in the North American region. For instance, in February 2022, Philips is supporting the American Heart Association's multi-year effort to generate awareness among cross-disciplinary specialties and improve survival rates from cardiovascular implantable electronic device (CIED) infections. Thus, owing to the abovementioned factors, the studied market in the North American region is expected to register significant growth over the forecast period.
Cardiovascular Devices Industry Overview
The cardiovascular devices market is moderately consolidated and competitive. The major players have established themselves in specific segments of the market. Furthermore, the companies are competing in emerging regions with global players and with established local players. Key players are developing and launching novel products and technologies to compete with existing products, while others are acquiring and partnering with other companies trending in the market. Some of the major players include Abbott, General Electric (GE Healthcare), W. L. Gore & Associates, Inc., Siemens Healthcare GmbH, Biotronik, Canon Medical Systems Corporation, B. Braun SE, LivaNova PLC, Boston Scientific Corporation, Cardinal Health, Edwards Lifesciences Corporation, and Medtronic.
Cardiovascular Devices Market Leaders
Boston Scientific Corporation
Abbott
Medtronic
Edwards Lifesciences Corporation
Cardinal Health
*Disclaimer: Major Players sorted in no particular order
Cardiovascular Devices Market News
- October 2022: Medtronic plc received United States FDA approval for expanded labeling of a cardiac lead that taps into the heart's natural electrical system, giving patients needed therapy while avoiding complications sometimes associated with traditional pacing methods, such as cardiomyopathy.
- October 2022 : Biosense Webster, Inc., part of Johnson & Johnson MedTech launched the HELIOSTAR Balloon Ablation Catheter in Europe. The HELIOSTAR Balloon Ablation Catheter is indicated for use in the catheter-based cardiac electrophysiological mapping (stimulating and recording) of the atria and, when used with a compatible multi-channel RF generator, for cardiac ablation.
Cardiovascular Devices Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Rapid Technological Advancements
4.2.2 Increasing Burden of Cardiovascular Diseases
4.2.3 Increased Preference for Minimally Invasive Procedures
4.3 Market Restraints
4.3.1 Stringent Regulatory Policies and Product Recalls
4.3.2 High Cost of Instruments and Procedures
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 Device Type
5.1.1 Diagnostic and Monitoring Devices
5.1.1.1 Electrocardiogram (ECG)
5.1.1.2 Remote Cardiac Monitoring
5.1.1.3 Other Diagnostic and Monitoring Devices
5.1.2 Therapeutic and Surgical Devices
5.1.2.1 Cardiac Assist Devices
5.1.2.2 Cardiac Rhythm Management Devices
5.1.2.3 Catheters
5.1.2.4 Grafts
5.1.2.5 Heart Valves
5.1.2.6 Stents
5.1.2.7 Other Therapeutic and Surgical Devices
5.2 Geography
5.2.1 North America
5.2.1.1 United States
5.2.1.2 Canada
5.2.1.3 Mexico
5.2.2 Europe
5.2.2.1 Germany
5.2.2.2 United Kingdom
5.2.2.3 France
5.2.2.4 Italy
5.2.2.5 Spain
5.2.2.6 Rest of Europe
5.2.3 Asia-Pacific
5.2.3.1 China
5.2.3.2 Japan
5.2.3.3 India
5.2.3.4 Australia
5.2.3.5 South Korea
5.2.3.6 Rest of Asia-Pacific
5.2.4 Middle East and Africa
5.2.4.1 GCC
5.2.4.2 South Africa
5.2.4.3 Rest of Middle East and Africa
5.2.5 South America
5.2.5.1 Brazil
5.2.5.2 Argentina
5.2.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Abbott
6.1.2 Boston Scientific Corporation
6.1.3 Cardinal Health
6.1.4 General Electric (GE Healthcare)
6.1.5 W. L. Gore & Associates, Inc.
6.1.6 Medtronic
6.1.7 Biotronik
6.1.8 Siemens Healthcare GmbH
6.1.9 Canon Medical Systems Corporation
6.1.10 Edwards Lifesciences Corporation
6.1.11 B. Braun SE
6.1.12 LivaNova PLC
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Cardiovascular Devices Industry Segmentation
As per the scope of the report, cardiovascular devices are used for the diagnosis of heart diseases and treatment of related health problems. The Cardiovascular Devices Market is Segmented by Device Type (Diagnostic and Monitoring Devices [Electrocardiogram (ECG), Remote Cardiac Monitoring, and Other Diagnostic and Monitoring Devices] and Therapeutic and Surgical Devices [Cardiac Assist Devices, Cardiac Rhythm Management Devices, Catheters, Grafts, Heart Valves, Stents, and Other Therapeutic and Surgical Devices]) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 different countries across major regions globally. The report offers the value (in USD million) for the above segments.
| Device Type | |||||||||
| |||||||||
|
| Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Cardiovascular Devices Market Research FAQs
How big is the Cardiovascular Devices Market?
The Cardiovascular Devices Market size is expected to reach USD 58.89 billion in 2024 and grow at a CAGR of 4.97% to reach USD 75.04 billion by 2029.
What is the current Cardiovascular Devices Market size?
In 2024, the Cardiovascular Devices Market size is expected to reach USD 58.89 billion.
Who are the key players in Cardiovascular Devices Market?
Boston Scientific Corporation, Abbott, Medtronic, Edwards Lifesciences Corporation and Cardinal Health are the major companies operating in the Cardiovascular Devices Market.
Which is the fastest growing region in Cardiovascular Devices Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Cardiovascular Devices Market?
In 2024, the North America accounts for the largest market share in Cardiovascular Devices Market.
What years does this Cardiovascular Devices Market cover, and what was the market size in 2023?
In 2023, the Cardiovascular Devices Market size was estimated at USD 56.10 billion. The report covers the Cardiovascular Devices Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Cardiovascular Devices Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
What are the biggest challenges for emerging market players in the Cardiovascular Device Industry?
The biggest challenges for emerging market players in the Cardiovascular Device Industry are a) Affordability concerns b) Limited access to advanced technologies c) Stringent regulatory frameworks
Cardiovascular Devices Industry Report
The global cardiovascular devices market is on an upward growth trajectory, propelled by the increasing prevalence of chronic heart diseases and the heightened demand for advanced diagnostic and surgical cardiology devices. Supported by a wave of drug approvals, clinical trials, and significant reductions in the prices of key therapeutic devices, these developments are enhancing accessibility in emerging markets. Robust R&D efforts are fostering innovations in product development, aiming to boost patient outcomes with advanced features like remote cardiac monitoring for enhanced safety and efficiency. Despite challenges such as complex regulatory frameworks, the market's potential for growth remains strong, driven by technological advancements and improved healthcare accessibility, indicating a promising outlook for the cardiovascular devices sector. Statistics for the Cardiovascular Devices market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Cardiovascular Devices analysis includes a market forecast outlook and historical overview. Get a sample of this industry analysis as a free report PDF download.
