Cancer Cachexia Market Size
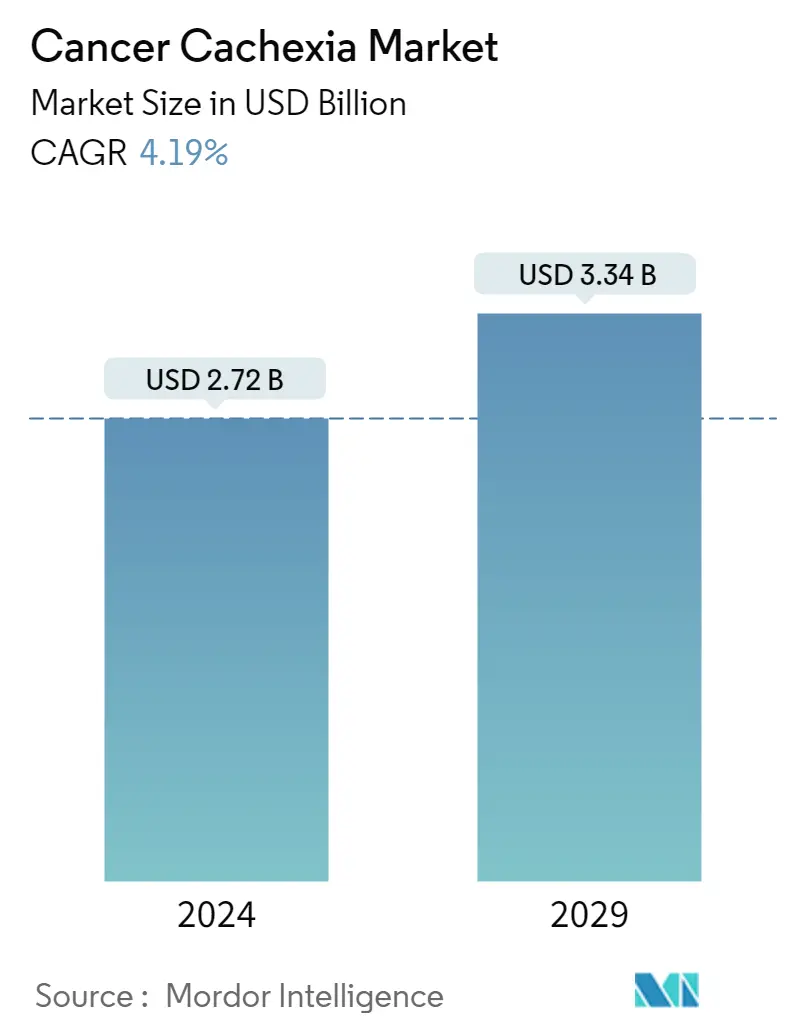
| Study Period | 2019 - 2029 |
| Market Size (2024) | USD 2.72 Billion |
| Market Size (2029) | USD 3.34 Billion |
| CAGR (2024 - 2029) | 4.19 % |
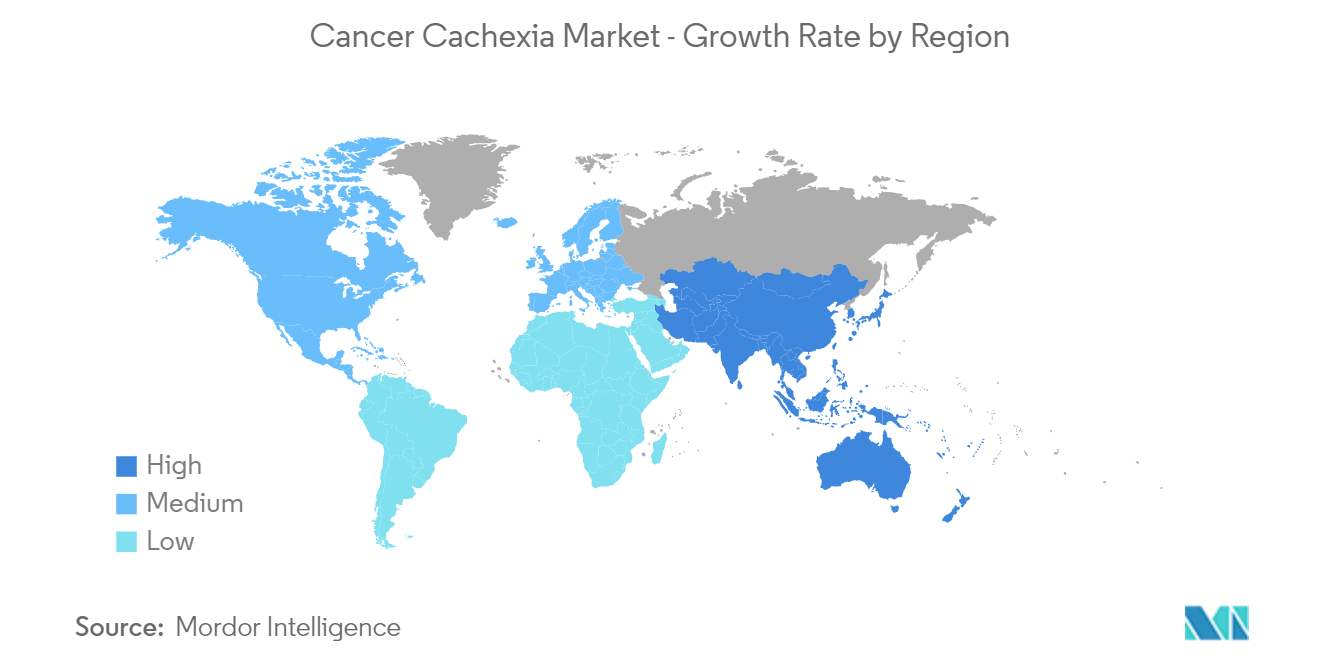
| Fastest Growing Market | Asia Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Cancer Cachexia Market Analysis
The Cancer Cachexia Market size is estimated at USD 2.72 billion in 2024, and is expected to reach USD 3.34 billion by 2029, growing at a CAGR of 4.19% during the forecast period (2024-2029).
The COVID-19 outbreak impacted the cancer cachexia market, as hospitals and healthcare services were significantly reduced due to social distancing measures enforced globally. The COVID-19 pandemic also affected the global economy and impacted general hospital care for non-COVID-19 patients in hospitals worldwide. According to a research article published in the British Journal of Nutrition, 2020, changes in nutritional status and weight loss during hospitalization are largely reported in some populations. However, it was not completely explored in COVID-19 patients. During the pandemic, the symptoms and associated conditions of COVID-19 deteriorated nutritional status, leading to cachexia. Cancer patients were at a higher risk of being infected with COVID-19, as they are immunocompromised, and their ability to fight against the infection was very low compared to other individuals. Additionally, if a cancer patient developed a COVID-19 infection, there were higher chances of developing multiple organ-related complications or cachexia. Hence, the COVID-19 pandemic affected the market significantly in it's preliminary phase, however currently the market is expected to have a favorable growth in the forecast period as the COVID-19 has subsided.
The increasing prevalence of cancer and cancer cachexia cases and the increasing investment in the R&D activities for developing novel therapeutics in the treatment of cancer cachexia are the major factors driving the market.
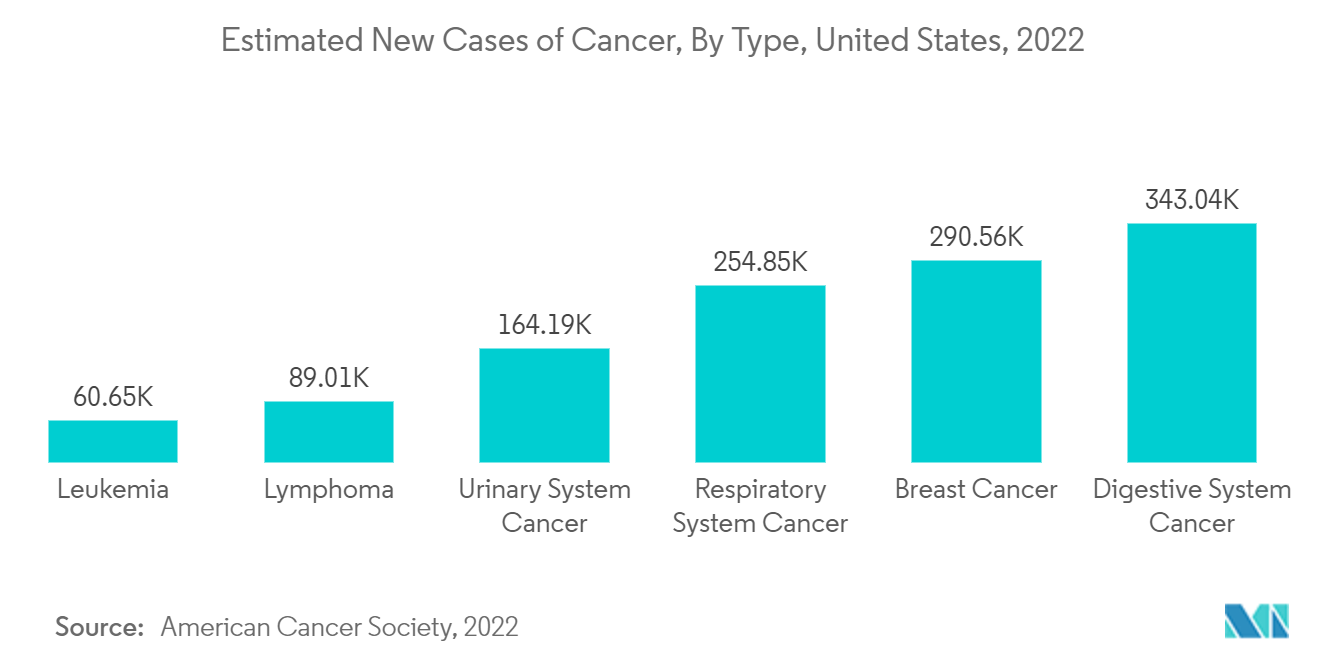
The global cancer burden is increasing. Thus, cancer therapies must be modified according to regional and national priorities. According to the International Agency for Research on Cancer (IARC) in December 2020, globally, 1 in 5 people develop cancer during their lifetime, and 1 in 8 men and 1 in 11 women die from the disease. According to the data published by WHO in February 2022, approximately 400,000 children develop cancer every year and the most common cancers vary between countries. Cervical cancer is the most common in 23 countries. Hence, the increasing burden of cancers worldwide also increases the prevalence of cancer cachexia among cancer patients, which is characterized by systemic inflammation, negative protein and energy balance, and an involuntary loss of lean body mass.
Due to this factor, most of the major players are focused on R&D activities to develop highly effective novel drugs in the treatment of cancer cachexia. This factor resulted in an increased pipeline of therapeutics waiting for approval and commercial launch post-approval. For instance, clinicaltrials.gov reported that, as of October 2022, a total of 150 studies were conducted or were being conducted to develop drugs that treat cancer cachexia. In December 2020, a clinical study was initiated by University of Copenhagen, in collaboration with Zealand University Hospital, the study was conducted to monitor the effect of medical cannabidiol on lean body mass (cachexia) in patients receiving Oxaliplatin- or Paclitaxel-based chemotherapy. Moreover, in January 2021, Helsinn Group and Ono Pharmaceutical Co. Ltd received the manufacturing and marketing approval of Adlumiz in Japan. It is a ghrelin receptor agonist to treat cancer cachexia in malignant tumors of non-small cell lung cancer, gastric cancer, pancreatic cancer, or colorectal cancer.Hence, the increasing research and development activities for cancer cachexia is expected to drive the growth of the market.
However, manufacturers of cancer cachexia drugs are discouraged from investing in the market because of the strict regulatory requirements and the long FDA approval procedure, which may impede the long-term growth of this market.
Cancer Cachexia Market Trends
This section covers the major market trends shaping the Cancer Cachexia Market according to our research experts:
Appetite Stimulators is Expected to Grow Significantly in the Cancer Cachexia Market
Cancer patients frequently develop loss of appetite and weight loss. Unintended weight loss and anorexia (the loss of appetite or desire to eat) in patients with cancer is associated with decreased performance status, reduced response and tolerance to treatment, decreased survival, and reduced quality of life. Therefore, the increasing use of an appetite stimulant could be considered for patients who exhibit decreased appetite. The increasing prevalence of cancer cachexia cases is one of the major factors propelling the overall market's growth.
According to an article published by PubMed in March 2022, pharmacologic interventions for cancer-related anorexia are very limited. International guidelines recommend promoting anabolism through appetite stimulants which help in nutritional, physical, and pharmacologic therapies. According to an article published by PubMed Central in August 2021, cachexia is a syndrome of metabolic disturbance characterised by inflammation and loss of muscle with or without loss of adipose tissue. In cancer cachexia, a multifaceted condition, patients suffer from loss of body weight and appetite that negatively impact the quality of life and survival of the patients. Thus, the high burden of cancer cachexia in quality of life of the patients is expected to increase the research and development activities regarding appetite stimulators for cancer cachexia, thus enhancing the segment growth.
With the growing burden of cancer worldwide, market players are investing steadily in innovative products and their R&D, thus boosting the market's growth. For instance, in March 2020, Pfizer Inc., a global pharmaceutical major, launched a global 12-week open-label study that will explore how its drug, PF-06946860, is tolerated in patients with cancer cachexia. Hence, such factors may help the appetite stimulators segment achieve a high growth rate in the future.
North America is Expected to Dominate the Cancer Cachexia Market
North America is expected to dominate the market due to the high burden of cancer and cancer cachexia, the presence of many drug manufacturers, and the advancing paradigm of care for cancer patients. According to the estimates of GLOBOCAN 2020, there are around 195,499 new cases of cancer in Mexico. Furthermore, according to the American Cancer Society 2022, there will be an estimated 1.9 million new cancer cases diagnosed in the United States in 2022.
Advancements in novel technology for the treatment of cancer cachexia and a strong product pipeline for wasting syndrome have also helped in the growth of the cancer cachexia market in the region. For instance, according to the data from clinicaltrials.gov in October 2022, a phase 1 clinical trial was started by the United States-based AVEO Pharmaceuticals, Inc. in February 2022, the study is designed to evaluate the safety, pharmacokinetics (PK), pharmacodynamics (PD), and immunogenicity in healthy subjects of a single dose of AV-380. AV-380 is an immunoglobulin (Ig) G1 monoclonal antibody (mAb) intended to bind circulating human growth differentiation factor 15 (GDF-15), a cytokine involved in cancer-induced cachexia. Hence, due to such factors, the market is expected to grow in the region.
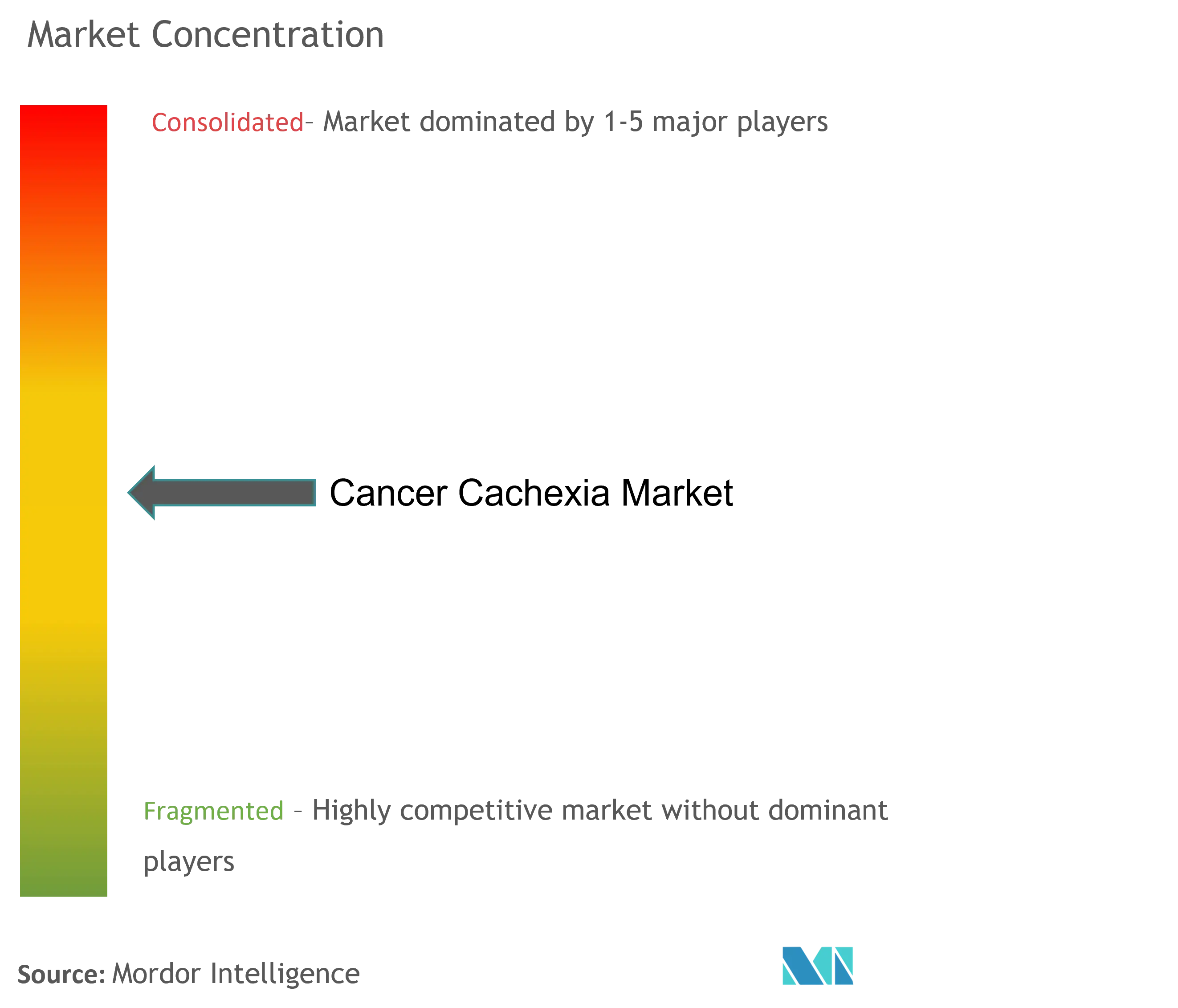
Cancer Cachexia Industry Overview
The cancer cachexia market is consolidated in nature due to the presence of very few major companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies which hold market shares and are well known. Key market players include Aeterna Zentaris, Aphios Corporation, Bristol-Myers Squibb Company, Pfizer Inc., Helsinn Group, Merck KGaA, Artelo Biosciences Inc., NGM Biopharmaceuticals, Aveo Oncology, Aavogen Inc., Green Cross Wellbeing Corporation, Cannabics Pharmaceuticals Inc., TCI Peptide Therapeutics, Fresenius Kabi, Tetra Bio-Pharma, and Actimed Therapeutics among others.
Cancer Cachexia Market Leaders
Merck & Co. Inc.
Aphios Corporation
Helsinn Group
Bristol-Myers Squibb Company
Pfizer
*Disclaimer: Major Players sorted in no particular order
Cancer Cachexia Market News
- August 2022: ARTHEx biotech launched ENTRYTM , a platform that will allow it to advance its pipeline of drugs that includes drugs for myotonic dystrophy, cancer cachexia, and osteoarthritis by providing tissue-enriched delivery of antimiRs.
- June 2022: The team led by Rutgers Cancer Institute of New Jersey received a USD 25 million Cancer Grand Challenges grant to research cancer cachexia, a debilitating condition people often experience in the later stages of their cancer.
Cancer Cachexia Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Deliverables
1.2 Study Assumptions
1.3 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increasing Prevalence of Cancer Cachexia
4.2.2 Strong R&D and Product Pipeline
4.3 Market Restraints
4.3.1 Stringent Regulatory Requirements
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Therapeutics
5.1.1 Progestogens
5.1.2 Corticosteroids
5.1.3 Combination Therapy
5.1.4 Other Therapeutics
5.2 By Mechanism of Action
5.2.1 Appetite Stimulators
5.2.2 Weight Loss Stabilizers
5.3 By Geography
5.3.1 North America
5.3.1.1 United States
5.3.1.2 Canada
5.3.1.3 Mexico
5.3.2 Europe
5.3.2.1 Germany
5.3.2.2 United Kingdom
5.3.2.3 France
5.3.2.4 Italy
5.3.2.5 Spain
5.3.2.6 Rest of Europe
5.3.3 Asia-Pacific
5.3.3.1 China
5.3.3.2 Japan
5.3.3.3 India
5.3.3.4 Australia
5.3.3.5 South Korea
5.3.3.6 Rest of Asia-Pacific
5.3.4 Middle-East and Africa
5.3.4.1 GCC
5.3.4.2 South Africa
5.3.4.3 Rest of Middle-East and Africa
5.3.5 South America
5.3.5.1 Brazil
5.3.5.2 Argentina
5.3.5.3 Rest of South America
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Aeterna Zentaris
6.1.2 Aphios Corporation
6.1.3 Bristol-Myers Squibb Company
6.1.4 Pfizer Inc.
6.1.5 Helsinn Group
6.1.6 Merck KGaA
6.1.7 Artelo Biosciences Inc.
6.1.8 NGM Biopharmaceuticals
6.1.9 Aveo Oncology
6.1.10 Aavogen Inc.
6.1.11 Green Cross Wellbeing Corporation
6.1.12 Cannabics Pharmaceuticals Inc.
6.1.13 TCI Peptide Therapeutics
6.1.14 Fresenius Kabi
6.1.15 Tetra Bio-Pharma
6.1.16 Actimed Therapeutics
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Cancer Cachexia Industry Segmentation
As per the scope of the report, cancer cachexia is a wasting syndrome characterized by weight loss, anorexia, asthenia, and anemia. The pathogenicity of this syndrome is multifactorial due to a complex interaction of tumor and host factors. The signs and symptoms of cachexia are considered as the prognostic parameters in cancer patients. Cancer cachexia drugs are used to treat or alleviate the condition. The cancer cachexia market is segmented by therapeutics (progestogen, corticosteroid, combination therapy, and other therapeutics), mechanism of action (appetite stimulators and weight loss stabilizers), and geography (North America, Europe, Asia-Pacific, Middle-East and Africa, and South America). The market report also covers the estimated market sizes and trends for 17 countries across major regions globally. The report offers the value (in USD million) for the above segments.
| By Therapeutics | |
| Progestogens | |
| Corticosteroids | |
| Combination Therapy | |
| Other Therapeutics |
| By Mechanism of Action | |
| Appetite Stimulators | |
| Weight Loss Stabilizers |
| By Geography | ||||||||
| ||||||||
| ||||||||
| ||||||||
| ||||||||
|
Cancer Cachexia Market Research FAQs
How big is the Cancer Cachexia Market?
The Cancer Cachexia Market size is expected to reach USD 2.72 billion in 2024 and grow at a CAGR of 4.19% to reach USD 3.34 billion by 2029.
What is the current Cancer Cachexia Market size?
In 2024, the Cancer Cachexia Market size is expected to reach USD 2.72 billion.
Who are the key players in Cancer Cachexia Market?
Merck & Co. Inc., Aphios Corporation, Helsinn Group, Bristol-Myers Squibb Company and Pfizer are the major companies operating in the Cancer Cachexia Market.
Which is the fastest growing region in Cancer Cachexia Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Cancer Cachexia Market?
In 2024, the North America accounts for the largest market share in Cancer Cachexia Market.
What years does this Cancer Cachexia Market cover, and what was the market size in 2023?
In 2023, the Cancer Cachexia Market size was estimated at USD 2.61 billion. The report covers the Cancer Cachexia Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Cancer Cachexia Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Cancer Cachexia Industry Report
Statistics for the 2024 Cancer Cachexia market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Cancer Cachexia analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
