Market Trends of Atherectomy Devices Industry
This section covers the major market trends shaping the Atherectomy Devices Market according to our research experts:
Directional Atherectomy Segment Holds a Significant Market Share in the Studied Market Over the Forecast Period
The directional atherectomy segment of the market is expected to witness significant growth in the forecast period over other products as it offers certain advantages such as low treatment cost, improved efficiency, etc. The increasing adoption of minimally invasive atherectomy procedures may boost the demand for atherectomy devices. According to a research study published by Krishna J. Rocha-Singh et. al., in June 2021, plaque removal with directional atherectomy (DA) before drug-coated balloon (DCB) angioplasty is a safe and effective treatment technique with a low provisional stent rate in patients with symptomatic severely calcified femoropopliteal artery disease. Thus, research studies like these are further expected to promote the usage of DA systems which is anticipated to fuel growth in the studied segment.
The strategic initiatives taken by key market players such as product launches, mergers and acquisitions, and partnerships are also expected to contribute to market growth. For instance, in August 2021, Medtronic submitted an application to the USFDA for the approval of the TurboHawk Plus Directional Atherectomy System, an updated version of its older and largely phased-out atherectomy device. This device will facilitate increased atherectomy procedures during Peripheral Arterial Disease (PAD) interventions. Additionally, in November 2021, Avinger Inc. received 510(k) approval from the United States FDA for the Pantherisimage-guided atherectomy system's new clinical indication. This clearance allows the company to directly market Pantheris forthe treatment of in-stent restenosis (ISR) in the lower extremity arteries.
Therefore, overall, with the growing number of surgeries, and product innovation, the directional atherectomy systems segment is expected to grow during the forecast period of the study.
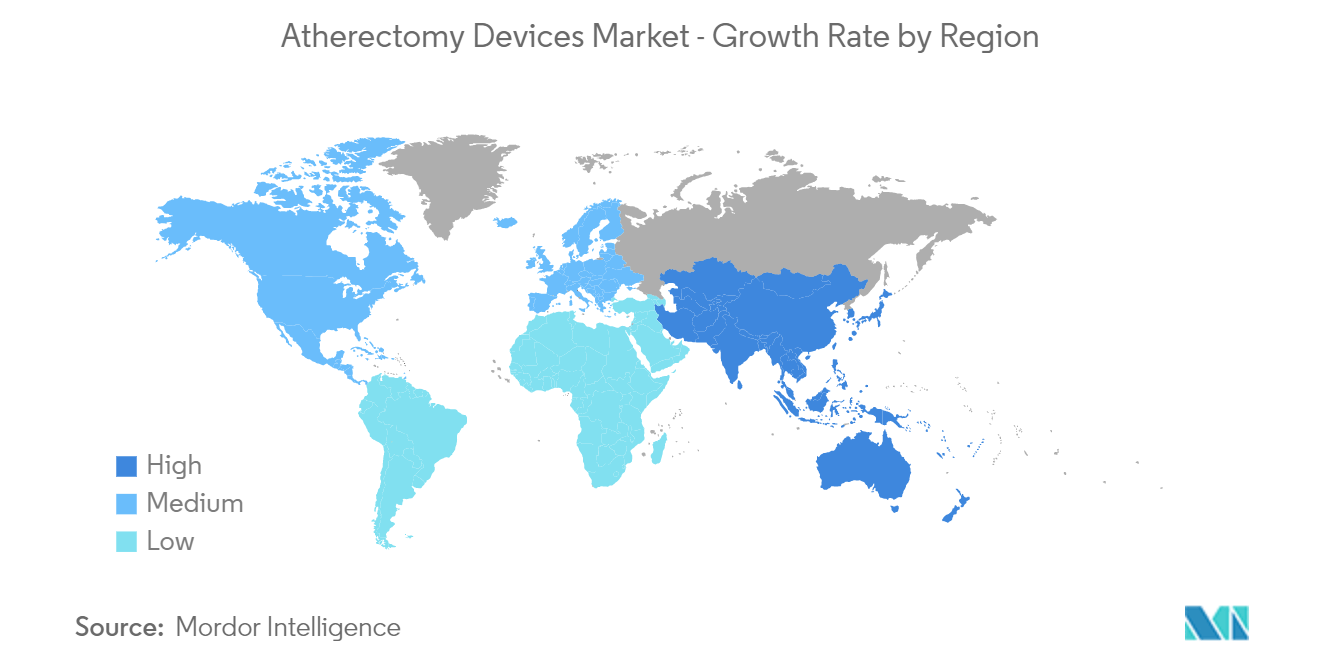
North America Holds a Significant Share in the Market and Expected to do Same during the Forecast Period
North America is expected to hold a significant share of the market. Factors such as increasing availability of reimbursements for atherectomy systems, adoption of atherectomy systems among medical professionals, the large patient population of peripheral and coronary artery diseases, and an increasing number of clinical trials (that aim to evaluate the therapeutic efficacy of atherectomy systems in specific disease treatment) are driving the growth of the North American atherectomy devices market.
According to the February 2022 update of the CDC, in the United States, heart diseases are the leading cause of death irrespective of gender, race, or ethnicity and the most common type of heart disease is coronary heart disease (CHD) or coronary artery disease in which coronary arteries become too narrow or cholesterol blockages develop in the walls, leading to stroke or heart attack. Thus, due to the rising prevalence of heart diseases, the demand for atherectomy procedures is expected to increase in the region which is anticipated to fuel growth in the studied market over the forecast period.
A suitable reimbursement scenario and a high number of FDA-approved devices are the key reasons for this dominance. For instance, in October 2021, BD (Becton, Dickinson and Company) announced it has received 510(k) clearance for expanded indications from the U.S. FDA for the Rotarex Atherectomy System. Additionally, in November 2021, Royal Philips announced the North American debut of new peripheral and coronary artery applications, including the IntraSight Mobile system and the Nexcimer laser system.
Thus, all above mentioned factors expected to boost the market in the region over the forecast period.