Asia-Pacific Hemodynamic Monitoring Market Size
| Study Period | 2021 - 2029 |
| Base Year For Estimation | 2023 |
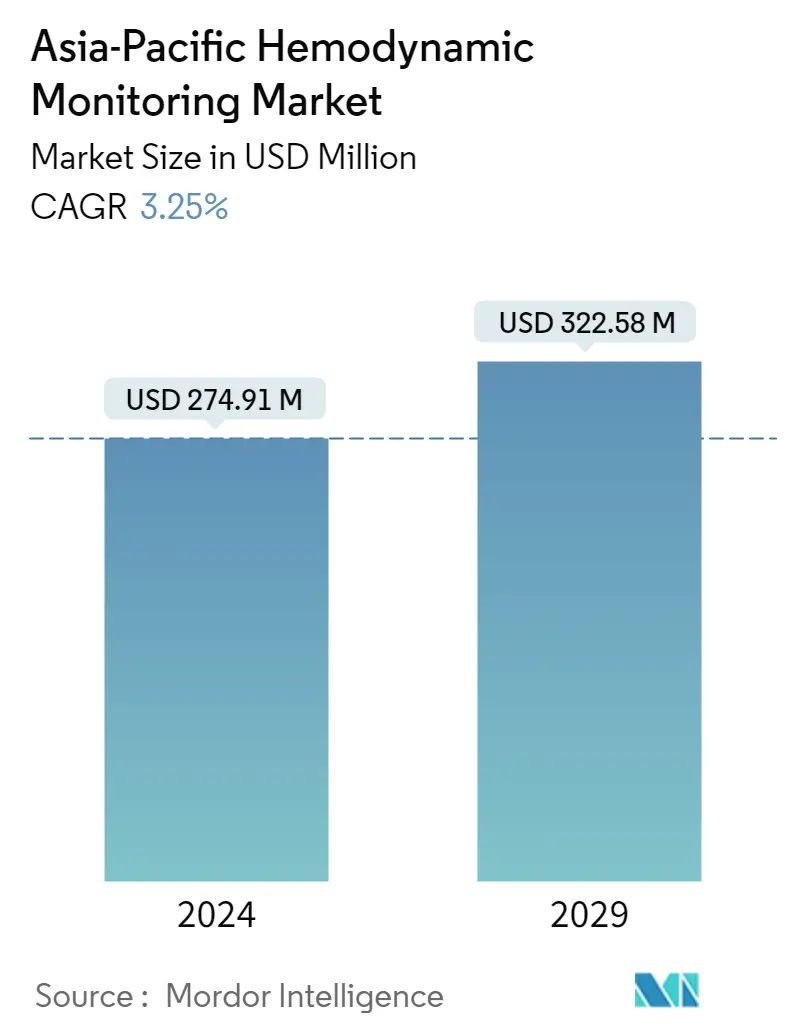
| Market Size (2024) | USD 274.91 Million |
| Market Size (2029) | USD 322.58 Million |
| CAGR (2024 - 2029) | 3.25 % |
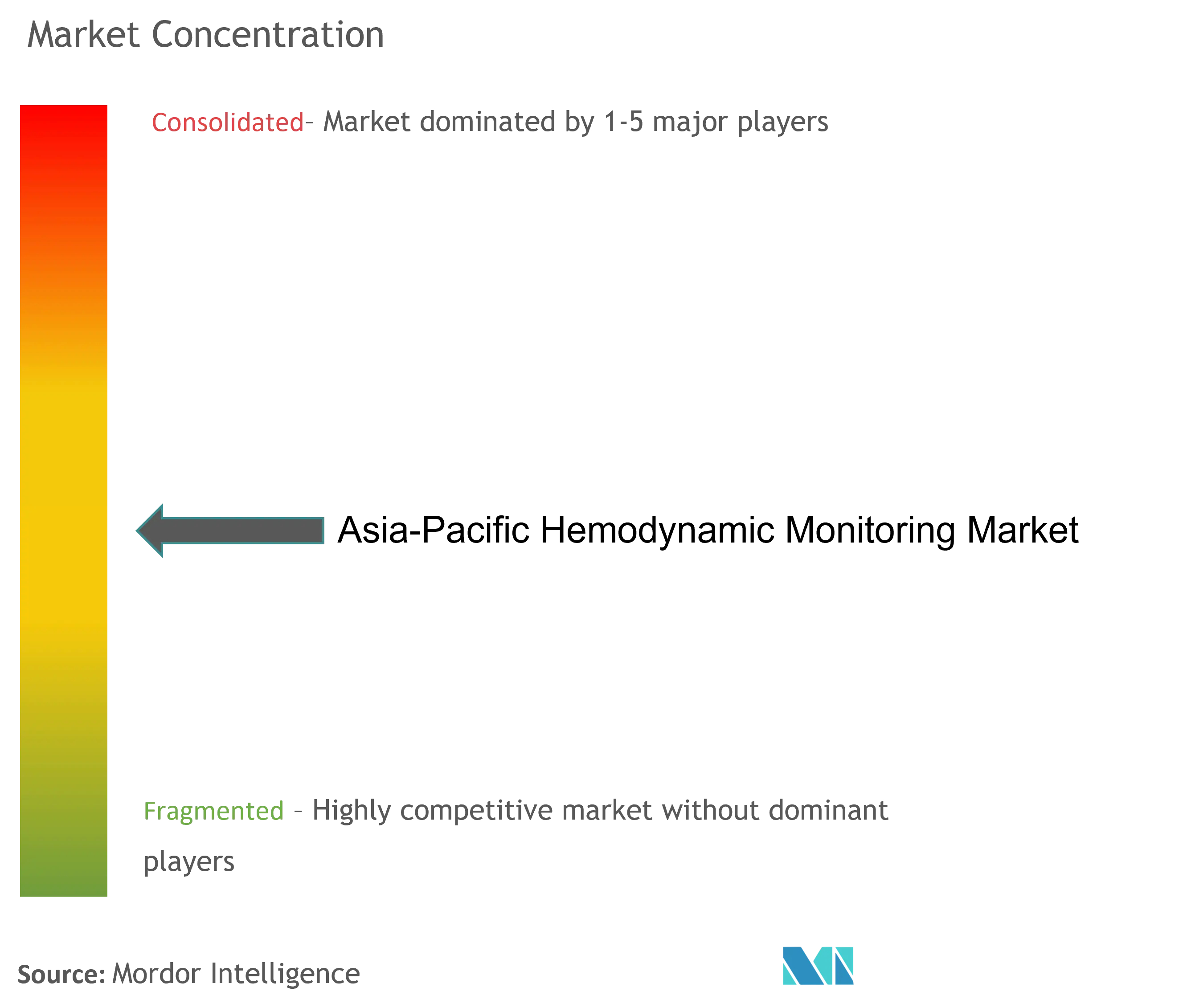
| Market Concentration | Medium |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Asia-Pacific Hemodynamic Monitoring Market Analysis
The Asia-Pacific Hemodynamic Monitoring Market size is estimated at USD 274.91 million in 2024, and is expected to reach USD 322.58 million by 2029, growing at a CAGR of 3.25% during the forecast period (2024-2029).
COVID-19 had a substantial market impact as the pandemic witnessed a sudden surge in the need for hemodynamic monitoring solutions for patients with SARS-CoV-2 infections. For instance, according to the LiDCO Group PLC's press release for 2021, demand for hemodynamic monitoring equipment increased from many existing customers, with significant short-term heightened demand from various hospitals in several countries, including a small number of monitors sold in Wuhan, China. Furthermore, some COVID-19 patients developed complications such as sepsis and required intensive care, wherein the healthcare settings were required to make use of advanced hemodynamic monitoring systems to improve patient outcomes. Hence, COVID-19 substantially boosted market demand for hemodynamic monitoring devices amid the pandemic. However, as per the analysis, the sudden heightened demand is likely to see a decline over the post-pandemic phase owing to the lesser number of COVID-19 patients and blood-related complications associated with these patients.
The key factors propelling the Asia-Pacific hemodynamic monitoring market are the increase in the critically ill geriatric population and the rising burden of various chronic diseases and cardiac disorders. According to OECD data updated in 2021, the percentage of the population aged 65 and over in China in 2020 was 11.97%. As a result, the demand for hemodynamic monitoring is projected to accelerate in the future, augmenting the growth of the studied market, as the elderly population is more susceptible to age-related blood disorders, such as hypertension, cardiovascular disease disorders, and cancers.
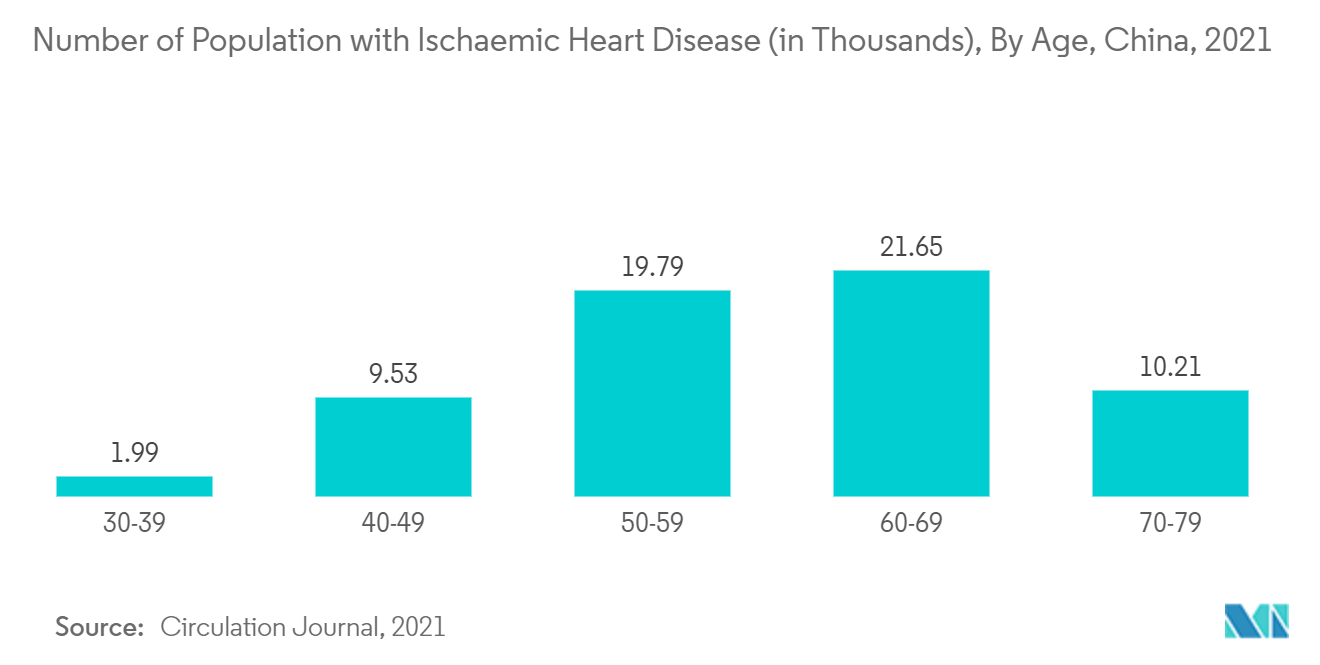
Insights on vital information such as blood volume, heart contractility, and fluid balance can be obtained through hemodynamic parameters in patients suffering from chronic diseases. all of which are required to assess the patient's general health and the efficacy of their treatment. The region is witnessing a surge in the prevalence and incidence of chronic disease over the past few years. For instance, the research article published in April 2022 in the International Journal of Public Health mentioned that CVD is one of the major healthcare burdens in China. According to the article, 290 million people are currently suffering from cardiac disorders, with stroke accounting for 13 million, coronary heart disease accounting for 11 million, rheumatic heart disease accounting for 2.5 million, heart failure accounting for 4.5 million, congenital heart disease accounting for 2 million, pulmonary heart disease accounting for 5 million, and hypertension accounting for 245 million. Moreover, out of the total mortality related to CVD, 40% occurs in China alone. Therefore, the increasing cases of CVDs are expected to increase the demand for hemodynamic monitoring systems, which is further expected to boost the growth of the Asia-Pacific hemodynamic monitoring market over the forecast period.
Therefore, owing to the aforementioned factors, it is anticipated that the studied market will witness growth over the analysis period. However, increased incidences of complications associated with invasive monitoring systems and stringent guidelines for the approval of new systems are expected to hinder the growth of the market.
Asia-Pacific Hemodynamic Monitoring Market Trends
This section covers the major market trends shaping the APAC Hemodynamic Monitoring Market according to our research experts:
Minimally Invasive Monitoring Systems is Expected to Hold High Market Share Over the Forecast Period
In the system segment of the market, minimally invasive monitoring systems are expected to have the largest market size and are expected to witness significant growth during the forecast period.
The minimally invasive systems help track stroke volume continuously and offer dynamic information on fluid responsiveness. Some systems assess volumetric preload variables, while others highlight the continuous measurement of central venous saturation with proprietary catheters. These variables of minimally invasive procedures and cardiac output deliver improved hemodynamic monitoring.
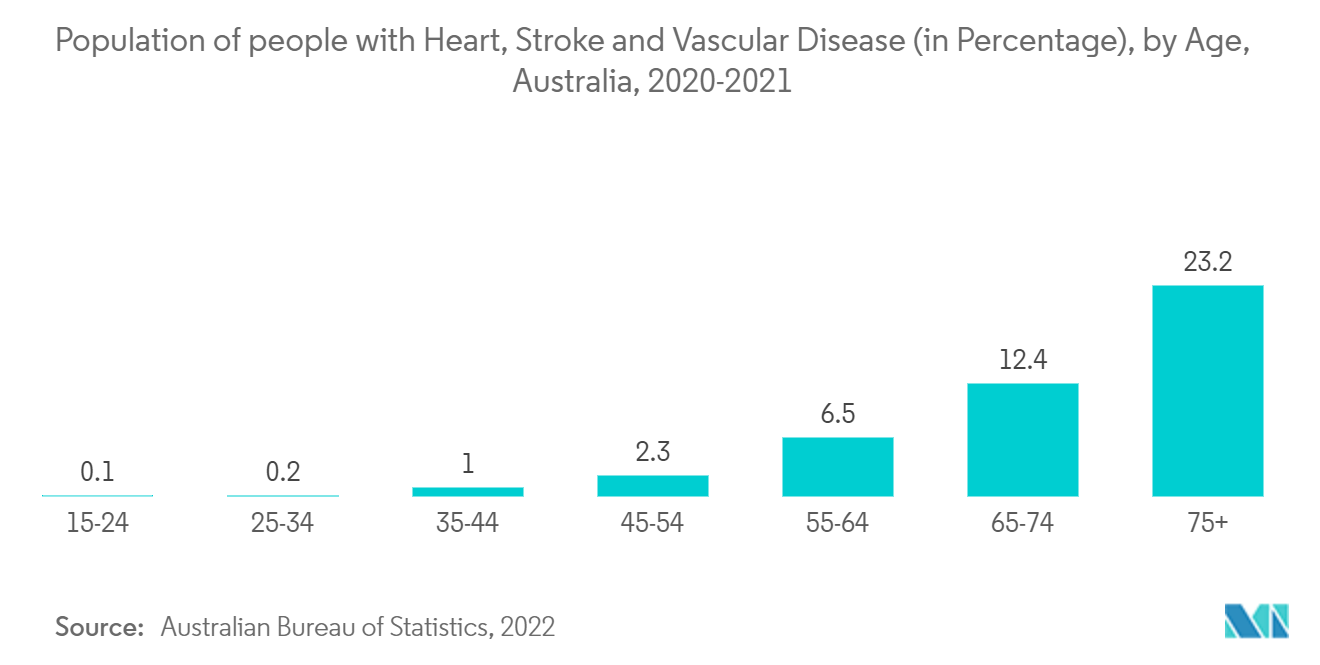
Moreover, there has been a recent advancement in echocardiography devices known as transesophageal echocardiography devices with a miniaturized probe, which are now used for continuous hemodynamic assessment. These monitoring systems have made hemodynamic calculations easier, gaining healthcare providers' attention and thus resulting in the segments' growth. Likewise, growth in the patient population is anticipated to have a positive impact on the segment's growth. For instance, as per Australian Bureau of Statistics data, in 2020-21, based on self-reported data from the National Health Survey, an estimated 571,000 Australians aged 18 and over (2.9% of the adult population) had coronary heart disease (CHD). With age, the frequency of CHD rises sharply, involving 1 in 9 (11%) persons aged 75 and above.
Thus, owing to the above-mentioned factors, the segment is expected to grow over the forecast period.
China is expected to dominate the Asia-Pacific Hemodynamic Monitoring Market Over the Forecast Period
China currently dominates the market for Asia-Pacific hemodynamic monitoring and is expected to continue its stronghold for a few more years. The growth of the hemodynamic monitoring market is most notable in China, owing to a significant number of market players, technologically advanced hospitals, an untapped patient population, and an increasing number of critically ill patients.
Congenital heart surgery (CHS) is frequently treated surgically and requires continuous cardiovascular and pulmonary monitoring for prompt identification and initiation of the treatment. According to the report released in May 2021 by Frontiers in Pediatrics, 61.3% of facilities performed fewer than 200 CHS cases, and 31.6% of all CHS cases were performed in limited facilities with a substantially lower volume than full-capacity facilities. Thus, for survival and the best clinical outcome, patients must have access to appropriate hospitals with congenital heart surgery (CHS) capabilities. As a result, it opens up attractive prospects for the major companies in the sector to enter the developing nation, which will help the market's overall expansion throughout the course of the study period. Therefore, such untapped opportunities are expected to increase the use of hemodynamic monitoring systems for cardiology applications within the country.
In addition, according to the China Cardiovascular Health and Disease Report 2021 released by the National Center for Cardiovascular Diseases, the number of people suffering from cardiovascular diseases in China is about 330 million. Thus, the growing incidence and prevalence of CVDs among the Chinese population will in turn boost the demand for effective monitoring of hemodynamic parameters during critical surgeries, ultimately bolstering the market demand for hemodynamic monitoring systems.
Therefore, such factors are expected to drive the growth of the market in the region in the future.
Asia-Pacific Hemodynamic Monitoring Industry Overview
The Asia-Pacific hemodynamic monitoring market is moderately competitive and consists of several major players, including Edwards Life Sciences Corporation, GE Healthcare, Getinge AB, ICU Medical Inc., and Koninklijke Philips NV, among others. In terms of market share, a few of the major players currently dominate the market. However, with technological advancements and product innovations, mid-size to smaller companies are increasing their market presence by introducing new products at lower prices.
Asia-Pacific Hemodynamic Monitoring Market Leaders
Edwards Life Sciences Corporation
GE Healthcare
Getinge AB
ICU Medical Inc.
Koninklijke Philips NV
*Disclaimer: Major Players sorted in no particular order
Asia-Pacific Hemodynamic Monitoring Market News
- In August 2022, a cooperative manufacturing agreement (CMA) was signed by CB Scientific, Inc., a designer, manufacturer, and distributor of non-invasive ambulatory cardiac monitoring products and services, and Shenzhen Pump Medical Co., Ltd. (Shenzhen Pump), a privately held, reputable medical device manufacturer, developer, and distributor of cardiovascular medical equipment and devices with headquarters in Shenzhen, Nanshan, China.
- In May 2021, Philips revealed the integration of its Interventional Hemodynamic System and the portable Patient Monitor IntelliVue X3, delivering advanced hemodynamic measures at the tableside in the cath lab and continuous monitoring of critical vital signs.
Asia-Pacific Hemodynamic Monitoring Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increase in the Critically-ill, Chronic Disease Patient Population
4.2.2 Technological Advancements by Medical Devices Companies
4.3 Market Restraints
4.3.1 Increased Incidences of Complications Associated with Invasive Monitoring Systems
4.3.2 Stringent Guidelines for Approval of New Systems
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products and Services
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By System
5.1.1 Minimally Invasive Monitoring Systems
5.1.2 Invasive Monitoring Systems
5.1.3 Non-invasive Monitoring Systems
5.2 By Application
5.2.1 Laboratory-based Monitoring Systems
5.2.2 Home-based Monitoring Systems
5.2.3 Hospital-based Monitoring Systems
5.3 Geography
5.3.1 China
5.3.2 Japan
5.3.3 India
5.3.4 Australia
5.3.5 South Korea
5.3.6 Rest of Asia-Pacific
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Edwards Life Sciences Corporation
6.1.2 LiDCO Group PLC
6.1.3 ICU Medical Inc.
6.1.4 Schwarzer Cardiotek GmbH
6.1.5 Koninklijke Philips NV
6.1.6 Medtronics (Cheetah Medical Inc.)
6.1.7 Getinge AB
6.1.8 GE Healthcare
6.1.9 Dragerwerk AG & Co. KGaA
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Asia-Pacific Hemodynamic Monitoring Industry Segmentation
As per the scope of the report, hemodynamic monitoring systems are used to measure the blood pressure inside the veins, heart, and arteries. These devices are also used to monitor the blood flow and the concentration of oxygen in the blood. The Asia-Pacific Hemodynamic Monitoring Market is segmented by System (Minimally Invasive Monitoring Systems, Invasive Monitoring Systems, and Non-invasive Monitoring Systems), Application (Laboratory-based, Home-based, and Hospital-based Monitoring Systems), and Geography (China, Japan, India, Australia, South Korea, and Rest of Asia-Pacific). The report offers the value (in USD million) for the above segments.
| By System | |
| Minimally Invasive Monitoring Systems | |
| Invasive Monitoring Systems | |
| Non-invasive Monitoring Systems |
| By Application | |
| Laboratory-based Monitoring Systems | |
| Home-based Monitoring Systems | |
| Hospital-based Monitoring Systems |
| Geography | |
| China | |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific |
Asia-Pacific Hemodynamic Monitoring Market Research FAQs
How big is the Asia-Pacific Hemodynamic Monitoring Market?
The Asia-Pacific Hemodynamic Monitoring Market size is expected to reach USD 274.91 million in 2024 and grow at a CAGR of 3.25% to reach USD 322.58 million by 2029.
What is the current Asia-Pacific Hemodynamic Monitoring Market size?
In 2024, the Asia-Pacific Hemodynamic Monitoring Market size is expected to reach USD 274.91 million.
Who are the key players in Asia-Pacific Hemodynamic Monitoring Market?
Edwards Life Sciences Corporation, GE Healthcare, Getinge AB, ICU Medical Inc. and Koninklijke Philips NV are the major companies operating in the Asia-Pacific Hemodynamic Monitoring Market.
What years does this Asia-Pacific Hemodynamic Monitoring Market cover, and what was the market size in 2023?
In 2023, the Asia-Pacific Hemodynamic Monitoring Market size was estimated at USD 266.26 million. The report covers the Asia-Pacific Hemodynamic Monitoring Market historical market size for years: 2021, 2022 and 2023. The report also forecasts the Asia-Pacific Hemodynamic Monitoring Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
APAC Non-invasive Monitoring Device Industry Report
Statistics for the 2024 APAC Non-invasive Monitoring Device market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. APAC Non-invasive Monitoring Device analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
