Asia-Pacific Electrophysiology Market Size
| Study Period | 2019 - 2029 |
| Base Year For Estimation | 2023 |
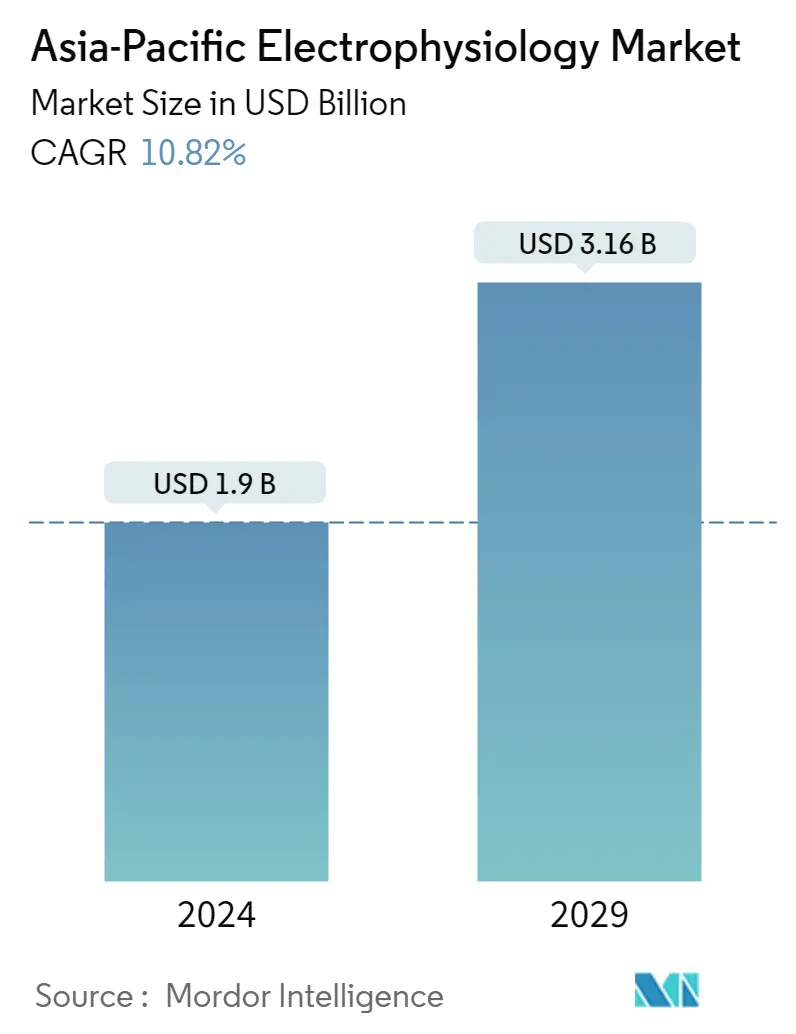
| Market Size (2024) | USD 1.90 Billion |
| Market Size (2029) | USD 3.16 Billion |
| CAGR (2024 - 2029) | 10.82 % |
| Market Concentration | High |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Asia-Pacific Electrophysiology Market Analysis
The Asia-Pacific Electrophysiology Market size is estimated at USD 1.9 billion in 2024, and is expected to reach USD 3.16 billion by 2029, growing at a CAGR of 10.82% during the forecast period (2024-2029).
The COVID-19 pandemic has had a substantial impact on the Asia-Pacific electrophysiology market initially. Most electrophysiology activities have been significantly reduced to accommodate the healthcare demands of the pandemic. For instance, according to the national center for biotechnology information (NCBI), in an article titled "Impact of COVID-19 pandemic on catheter ablation in China: A spatiotemporal analysis," published in 2022, catheter ablation increased remarkably from around 100,019 in 2020 to 125,006 in 2021. The catheter experienced a substantial decline during the early phase of the COVID-19 pandemic and then gradually returned to pre-COVID-19 levels. Thus, the COVID-19 outbreak affected the market's growth adversely in its preliminary phase. However, the market is expected to gain traction due to the significant increase in electrophysiology activities.
Further, the increasing prevalence of targeted diseases and technological advancements in the field of electrophysiology are among the major factors driving the growth of the studied market. For instance, according to the National Library for Biotechnology Information, in an article titled "Cardiac electrophysiology in India: Academic publications in the last decade and the way forward," in 2021, it has been stated that during the past twenty-five years, the field of cardiac electrophysiology witnessed a steady rise, in parallel to the developments happening all around the world. Thereby, due to such factors, the market is expected to have significant growth during the forecast period.
Additionally, according to the Organisation for Economic Cooperation and Development (OECD), in an article titled "Health at a Glance: Asia/Pacific 2022," cardiovascular disease (CVD) is the leading cause of death in Asia-Pacific. Thereby, a high prevalence of heart diseases may elevate the need for electrophysiology.
Moreover, in 2021, Stereotaxis and Shanghai Microport EP Medtech Co., Ltd. mentioned that they have collaborated to advance technology innovation and commercial adoption of robotics in electrophysiology in China. The agreement brings together MicroPort EP's commercial and product leadership in China's electrophysiology market with Stereotaxis advanced Robotic Magnetic Navigation technology. Thereby, such product expansions and collaborations is elevating the growth og the electrophysiology market within the region.
Therefore, owing to the aforementioned factors the studied market is anticipated to witness growth over the analysis period. However, the lack of skilled professionals is likely to impede market growth.
Asia-Pacific Electrophysiology Market Trends
Atrial Fibrillation Expected to Hold Significant Market Share
Atrial fibrillation (AF) is a common abnormal heart rhythm that occurs when electrical impulses fire off from different places in the atria in a disorganized way. It often begins as short periods of abnormal beating, which become longer or continuous over time. High blood pressure and valvular heart disease are the most common modifiable risk factors for AF. Atrial fibrillation is one of the most common types of cardiac arrhythmia among adults.
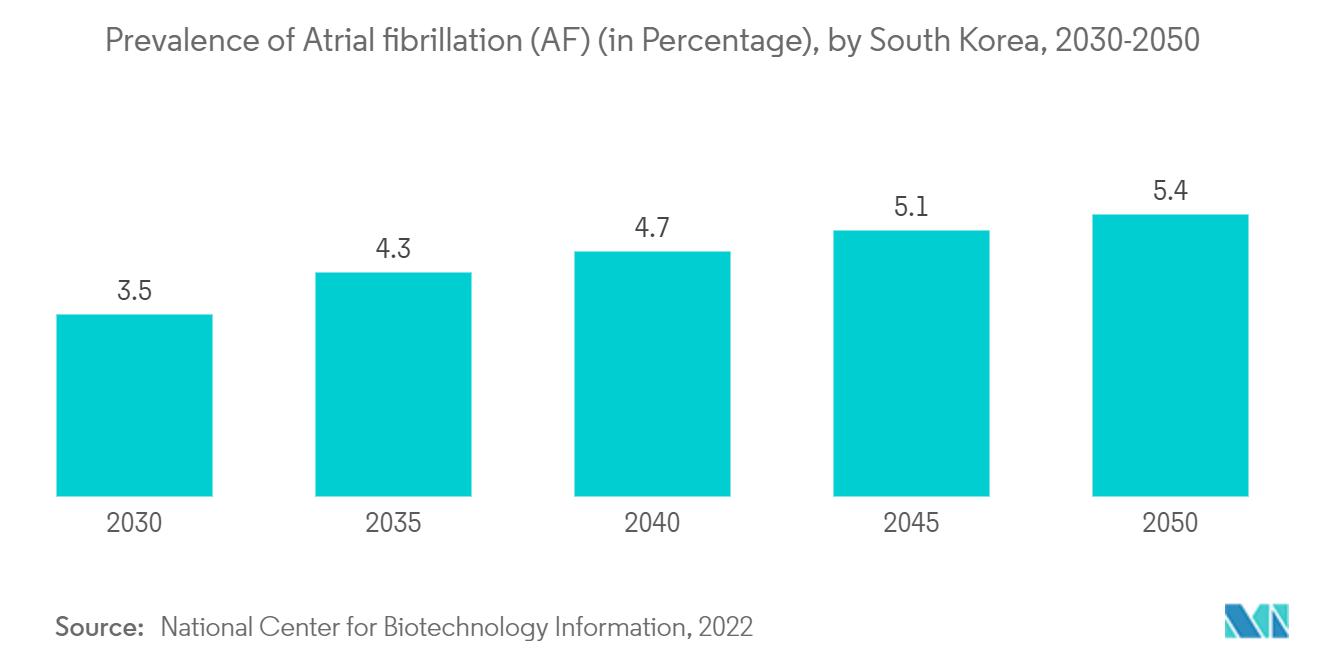
For instance, it was stated by the NCBI in 2022 that atrial fibrillation (AF) is a global problem, with an increasing incidence and prevalence with an aging population. Similar to Western countries, the prevalence rates of AF will continuously increase in the following decades in the Asia Pacific region. Thereby, the prevalence of atrial fibrillation within the region is expected to elevate the segment's growth.
Additionally, according to an article titled "Atrial Fibrillation: Improving care pathways to meet the rising burden across the Asia-Pacific region," in 2022, the prevalence of Atrial fibrillation (AFib) in the Asia-Pacific region has increased in recent decades, and this is set to continue increasing. A similar source stated that, by 2050, around 49 million men and 23 million women in the Asia Pacific will carry a diagnosis of AF. Thereby, the increasing prevalence of atrial fibrillation is going to elevate the growth of the market due to higher need for electrophysiology.
Therefore, the atrial fibrillation segment is expected to witness significant growth over the forecast period due to the abovementioned factors.
China Expected to Witness Healthy Growth
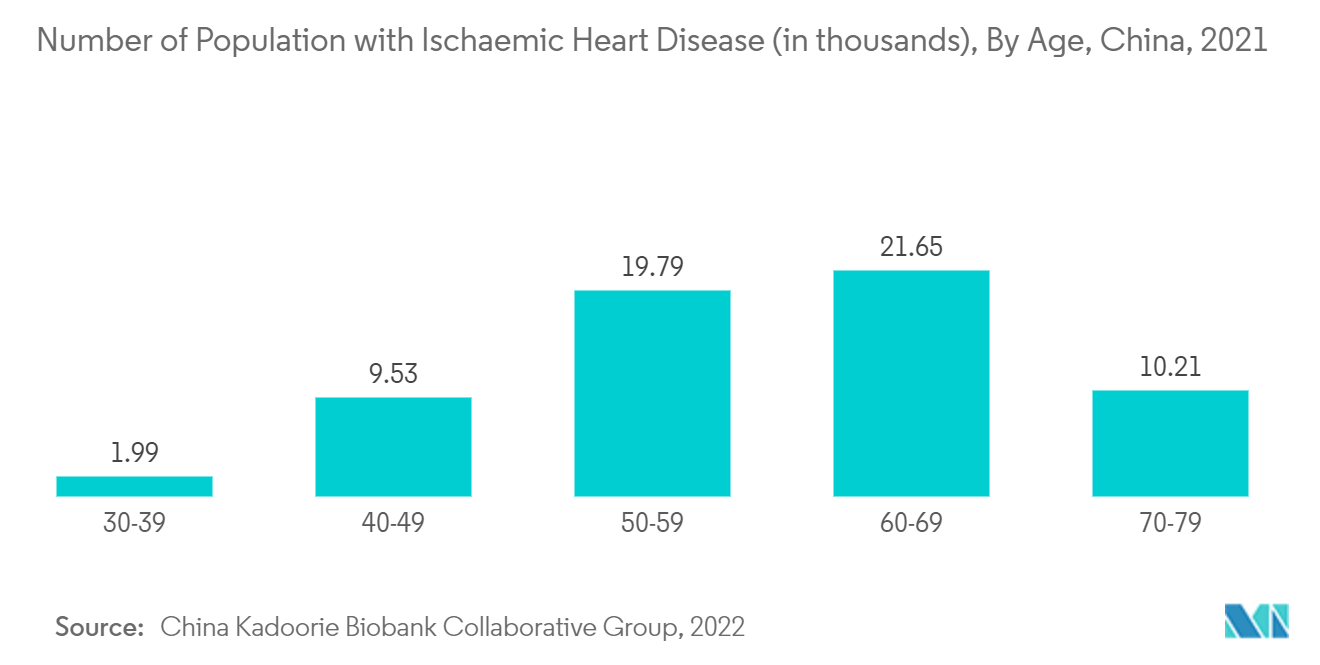
China is expected to witness healthy growth in the market owing to factors such as the rise in the number of arrhythmia and heart failure cases in the country.
In addition, according to an article published by the Journal of Arrhythmia, titled "Asia Pacific Heart Rhythm Society (APHRS) practice guidance on atrial fibrillation screening," in 2021, atrial fibrillation is the most common sustained arrhythmia globally, and the prevalence increased by twenty times over a period of 11 years in China. Thereby, the prevalence of atrial fibrillation in China is elevating the growth of the market in the country.
According to the NCBI, in an article titled "Cardiovascular Disease Mortality and Potential Risk Factor in China: A Multi-Dimensional Assessment by a Grey Relational Approach," in 2022, cardiovascular disease was the leading cause of death globally, and China represented 40% of deaths due to cardiovascular diseases. Thereby, there is a higher need for various diagnostics in order to get cardiovascular diseases identified. This may elevate the growth of the market.
Asia-Pacific Electrophysiology Industry Overview
The Asia-Pacific electrophysiology market is highly competitive in nature due to the presence of several companies operating globally and regionally. The competitive landscape section of the report includes an analysis of some international and local companies that hold significant market shares and are well known, including Abbott Laboratories, Boston Scientific Corporation, and Medtronic.
Asia-Pacific Electrophysiology Market Leaders
Abbott Laboratories
Boston Scientific Corporation
GE Healthcare
Medtronic PLC
Koninklijke Philips N.V.
*Disclaimer: Major Players sorted in no particular order
Asia-Pacific Electrophysiology Market News
- August 2021: Stereotaxis and Shanghai Microport EP Medtech Co. Ltd announced a broad collaboration to advance technology innovation and commercial adoption of robotics in electrophysiology in China.
- June 2021: CardioFocus stated that it had entered a licensing and distribution agreement with China Grand Pharmaceutical to seek regulatory approvals for the HeartLight X3 catheter ablation technology in China. The HeartLight X3 system is used for providing controlled and consistent pulmonary vein isolation, which is the primary treatment for atrial fibrillation (AFib).
APAC Electrophysiology Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Overview
4.2 Market Drivers
4.2.1 Increasing Prevalence of Targeted Diseases
4.2.2 Technological Advancements in the Field of Electrophysiology
4.3 Market Restraints
4.3.1 Lack of Skilled Professionals
4.4 Porter's Five Forces Analysis
4.4.1 Threat of New Entrants
4.4.2 Bargaining Power of Buyers/Consumers
4.4.3 Bargaining Power of Suppliers
4.4.4 Threat of Substitute Products
4.4.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION (Market Size by Value - USD million)
5.1 By Product
5.1.1 Ablation Catheters
5.1.2 Laboratory Devices
5.1.3 Diagnostic Catheters
5.1.4 Access Devices
5.1.5 Other Products
5.2 By Target Disease
5.2.1 Atrial Fibrillation
5.2.2 Atrial Flutter
5.2.3 Wolff-Parkinson-White Syndrome
5.2.4 Atrioventricular Nodal Reentry Tachycardia
5.2.5 Other Diseases
5.3 Geography
5.3.1 China
5.3.2 Japan
5.3.3 India
5.3.4 Australia
5.3.5 South Korea
5.3.6 Rest of Asia-Pacific
6. COMPETITIVE LANDSCAPE
6.1 Company Profiles
6.1.1 Abbott Laboratories
6.1.2 Biotronik SE & Co. KG
6.1.3 Boston Scientific Corporation
6.1.4 GE Healthcare
6.1.5 Johnson & Johnson
6.1.6 Medtronic PLC
6.1.7 Microport Scientific Corporation
6.1.8 Koninklijke Philips N.V.
6.1.9 Siemens Healthineers
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Asia-Pacific Electrophysiology Industry Segmentation
As per the scope of the report, electrophysiology is a biomedical field that deals with the study of electric activity in the body. Electrophysiology includes the study of the generation of electrical activity and its effects on the body.
The Asia-Pacific electrophysiology market is segmented by Product (Ablation Catheters, Laboratory Devices, Diagnostic Catheters, Access Devices, and Other Products), Target Disease (Atrial Fibrillation, Atrial Flutter, Wolff-Parkinson-White Syndrome, Atrioventricular Nodal Reentry Tachycardia and Other Target Diseases) and Geography (China, Japan, India, Australia, South Korea and Rest of Asia-Pacific). The report offers the value in USD million for the above segments.
| By Product | |
| Ablation Catheters | |
| Laboratory Devices | |
| Diagnostic Catheters | |
| Access Devices | |
| Other Products |
| By Target Disease | |
| Atrial Fibrillation | |
| Atrial Flutter | |
| Wolff-Parkinson-White Syndrome | |
| Atrioventricular Nodal Reentry Tachycardia | |
| Other Diseases |
| Geography | |
| China | |
| Japan | |
| India | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific |
APAC Electrophysiology Market Research FAQs
How big is the Asia-Pacific Electrophysiology Market?
The Asia-Pacific Electrophysiology Market size is expected to reach USD 1.90 billion in 2024 and grow at a CAGR of 10.82% to reach USD 3.16 billion by 2029.
What is the current Asia-Pacific Electrophysiology Market size?
In 2024, the Asia-Pacific Electrophysiology Market size is expected to reach USD 1.90 billion.
Who are the key players in Asia-Pacific Electrophysiology Market?
Abbott Laboratories, Boston Scientific Corporation, GE Healthcare, Medtronic PLC and Koninklijke Philips N.V. are the major companies operating in the Asia-Pacific Electrophysiology Market.
What years does this Asia-Pacific Electrophysiology Market cover, and what was the market size in 2023?
In 2023, the Asia-Pacific Electrophysiology Market size was estimated at USD 1.71 billion. The report covers the Asia-Pacific Electrophysiology Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Asia-Pacific Electrophysiology Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Asia-Pacific Electrophysiology Industry Report
Statistics for the 2024 Asia-Pacific Electrophysiology market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Asia-Pacific Electrophysiology analysis includes a market forecast outlook to for 2024 to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
