Alcohol Ingredients Market Size
| Study Period | 2019 - 2029 |
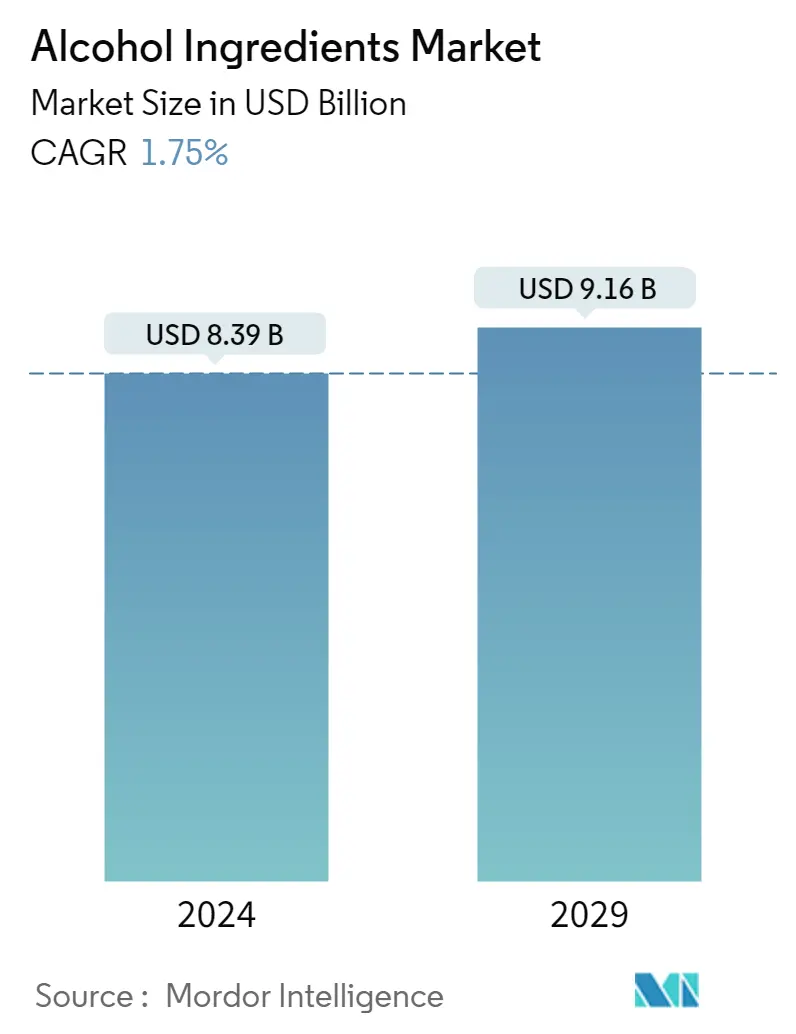
| Market Size (2024) | USD 8.39 Billion |
| Market Size (2029) | USD 9.16 Billion |
| CAGR (2024 - 2029) | 1.75 % |
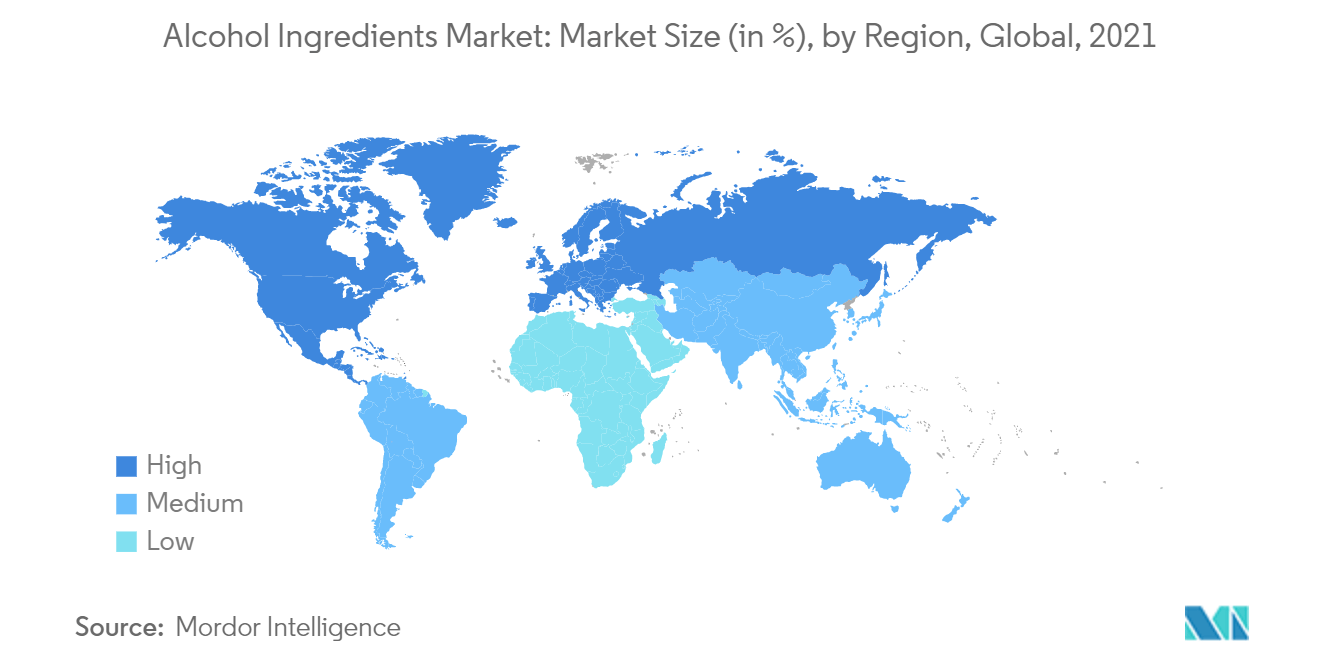
| Fastest Growing Market | Asia Pacific |
| Largest Market | Europe |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Alcohol Ingredients Market Analysis
The Alcohol Ingredients Market size is estimated at USD 8.39 billion in 2024, and is expected to reach USD 9.16 billion by 2029, growing at a CAGR of 1.75% during the forecast period (2024-2029).
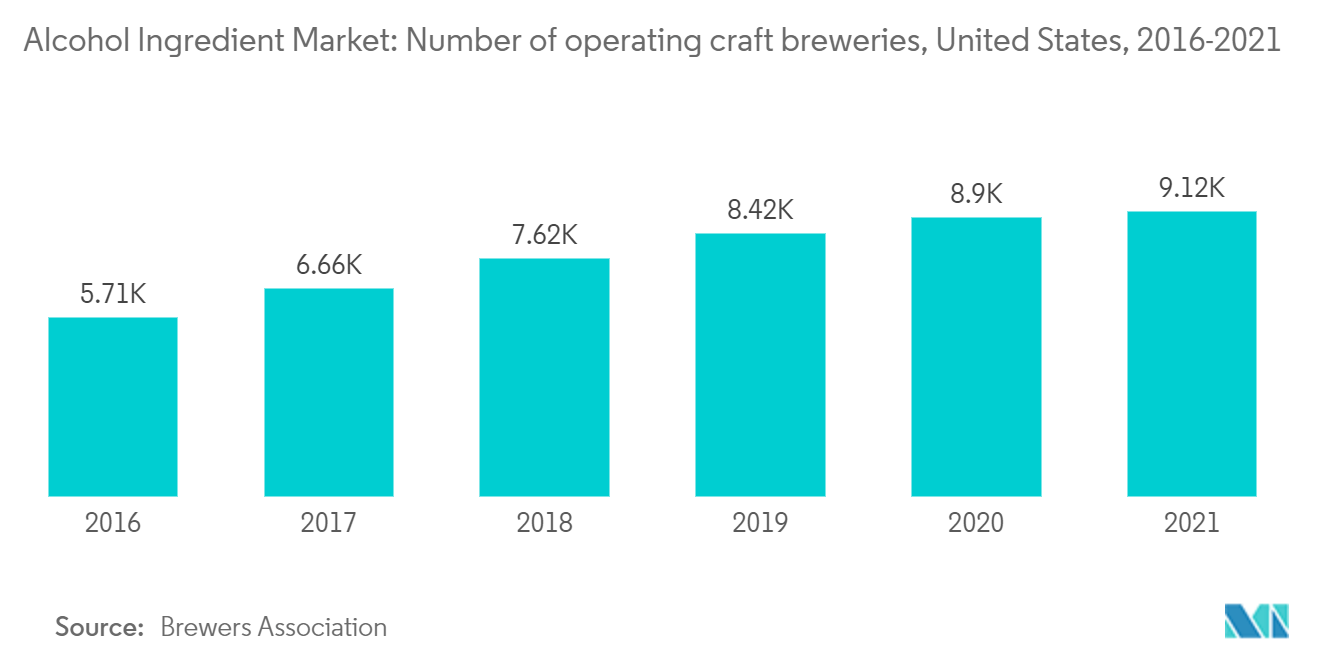
According to Lucas Bols's annual reports, in recent years, alcohol consumption has increased significantly around the globe. Bols, a Dutch public company specializing in producing, distributing, selling, and marketing alcoholic beverages, reported a revenue increase in Western Europe from EUR 34.4 million in 2016 to EUR 42.11 million in 2021. But the company's revenue dropped to EUR 30.75 million as a result of COVID-19. However, post-pandemic, the market has recovered, and ingredient manufacturers are increasing their production to meet the burgeoning demand coming from the alcoholic beverage sector. Moreover, consumers are increasingly inclined toward craft beer, which has resulted in an increasing number of craft breweries around the globe, with the highest number of outlets in the United Kingdom, followed by Germany, Italy, and others.
This is yet another factor driving the alcohol ingredients market around the globe. Additionally, manufacturers are increasing their production capacity owing to the rising demand. For instance, in August 2021, Angel Yeast invested heavily in acquisitions and production. Together with Shandong Lufa Holding company, a local investment corporation within the Chinese mainland, the company announced the acquisition of Bio Sunkeen for RMB 60 million (USD 9.2 million). Additionally, manufacturers are innovating various types of ingredients colorants, yeast, flavors & salt, enzymes, and others to enhance the quality of alcoholic beverages.
Alcohol Ingredients Market Trends
This section covers the major market trends shaping the Alcohol Ingredients Market according to our research experts:
Increasing Demand For Craft Beer
The demand for the craft brewing and distilling industries experienced significant growth during the past few years. Consumers across the world are seeking distinctive and flavored beers. This is pushing the alcohol ingredients market into the beer market space. The brewing industry utilizes five major ingredients, namely malt ingredient extract adjuncts/grains, hops, beer yeast, and beer additives. A mixture of starch sources may be used, with a secondary saccharide, such as maize (corn), rice, or sugar, as these are often termed adjuncts, especially when used as a lower-cost substitute for malted barley. Less widely used starch sources include millet, sorghum, and cassava root in Africa, potato in Brazil, and agave in Mexico, among others. Brewing-grade malt extracts are made with the highest-quality brewing malts and get additional colors and flavors from using specialty malts. This gives beer the unique character and flavor desired for the particular style of brew. These malts often have a longer time in the kiln, at higher temperatures, or get roasted to add depth and complexity to the resulting beer. Moreover, the increasing development of microbreweries and the growing production of craft beer led to a growing demand for craft beers.
Asia-Pacific Remains the Fastest Growing Region
Asia-Pacific, in particular, holds immense potential for the craft beer market, with robust demand for Westernized craft beer that is blended with local tastes and preferences. Shift in consumer preferences from traditional alcohol toward imported alcohol has been one of the driving factors for the growth of the alcoholic beverage market in the region. For instance, according to the Ministry of Agriculture, Forestry, and Fisheries, in 2021, approximately 188 billion Japanese Yen of wine was imported to Japan, making up most of the imports. Despite the fact that fruit wine was produced in various prefectures in Japan, imports covered the majority of domestic demand. In countries like China, consumers are inclined toward wines manufactured by famous vineyard regions, such as Bordeaux, owing to renowned heritage and prestige. Whiskies, Cognacs, and imported wines are becoming popular across the region as consumers are more inclined toward premium products.
Alcohol Ingredients Industry Overview
The global alcohol ingredient market is highly fragmented due to the dominance of several local and global players present in the market. Nonetheless, the market exhibits a strong presence of global key players, such as DSM, ADM, Cargill, and Döhler Group, alongside other domestic producers. Expansion remained the most adopted strategy among malt ingredient manufacturers, holding a major chunk of total strategies, followed by product innovation, partnership, and merger and acquisition. The major players are expanding their production capacities and strategically planning to supplement their export facilities in order to stand out among other manufacturers in the market.
Alcohol Ingredients Market Leaders
Kerry Group plc
Cargill, Incorporated
Archer Daniels Midland Company
Dohler Group SE
Koninklijke Dsm N.V
*Disclaimer: Major Players sorted in no particular order
Alcohol Ingredients Market News
- December 2021: Givaudan acquired DDW, the Color House, a US-based natural color company. The acquisition of DDW further strengthened Givaudan's leadership position in the color business globally.
- December 2021: The AEB Group launched FERMOALE New-E, a new yeast in active dry form suitable to produce juicy beer styles.
- September 2021: Archer-Daniels-Midland Co., a grain merchant from the United States, announced that it opened a new flavor production factory in China to manufacture food and beverages in response to the growing consumer demand.
Alcohol Ingredients Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions and Market Definition
1.2 Scope of the Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET DYNAMICS
4.1 Market Drivers
4.2 Market Restraints
4.3 Porter's Five Forces Analysis
4.3.1 Threat of New Entrants
4.3.2 Bargaining Power of Buyers/Consumers
4.3.3 Bargaining Power of Suppliers
4.3.4 Threat of Substitute Products
4.3.5 Intensity of Competitive Rivalry
5. MARKET SEGMENTATION
5.1 Ingredient Type
5.1.1 Yeast
5.1.2 Enzymes
5.1.3 Colorants
5.1.4 Flavors & Salts
5.2 Beverage Type
5.2.1 Beer
5.2.2 Spirits
5.2.3 Wine
5.2.4 Whisky
5.2.5 Brandy
5.3 Geography
5.3.1 North America
5.3.1.1 United States
5.3.1.2 Canada
5.3.1.3 Mexico
5.3.1.4 Rest of North America
5.3.2 Europe
5.3.2.1 Spain
5.3.2.2 United Kingdom
5.3.2.3 Germany
5.3.2.4 France
5.3.2.5 Italy
5.3.2.6 Russia
5.3.2.7 Rest of Europe
5.3.3 Asia-Pacific
5.3.3.1 China
5.3.3.2 Japan
5.3.3.3 India
5.3.3.4 Australia
5.3.3.5 Rest of Asia-Pacific
5.3.4 South America
5.3.4.1 Brazil
5.3.4.2 Argentina
5.3.4.3 Rest of South America
5.3.5 Middle East and Africa
5.3.5.1 South Africa
5.3.5.2 United Arab Emirates
5.3.5.3 Rest of Middle East and Africa
6. COMPETITIVE LANDSCAPE
6.1 Most Adopted Strategies
6.2 Market Share Analysis
6.3 Company Profiles
6.3.1 Cargill, Incorporated
6.3.2 Archer Daniels Midland Company
6.3.3 Ashland Inc.
6.3.4 D.D. Williamson & Co. Inc.
6.3.5 Koninklijke Dsm NV
6.3.6 Kerry Group PLC
6.3.7 Treatt PLC
6.3.8 Chr Hansen Holdings A/S
6.3.9 Sensient Technologies Corporation
6.3.10 Dohler Group
- *List Not Exhaustive
7. MARKET OPPORTUNITIES AND FUTURE TRENDS
Alcohol Ingredients Industry Segmentation
Alcohol ingredients are added to grains, fruits, or vegetables, which are fermented with yeast, enzymes, color, flavor, and salt as part of the production process. The global alcohol ingredients market (henceforth, referred to as the market studied) is segmented by ingredients type, beverage type, and geography. By ingredient type, the market is segmented into yeast, enzymes, colorants, flavors, and salts. By beverage type, the market is segmented into beer, wine, brandy, whiskey, and spirits. It provides an analysis of emerging and established economies across the world, comprising North America, Europe, South America, Asia-Pacific, and Middle East and Africa. For each segment, the market sizing and forecasts have been provided on the basis of value (in USD million).
| Ingredient Type | |
| Yeast | |
| Enzymes | |
| Colorants | |
| Flavors & Salts |
| Beverage Type | |
| Beer | |
| Spirits | |
| Wine | |
| Whisky | |
| Brandy |
| Geography | |||||||||
| |||||||||
| |||||||||
| |||||||||
| |||||||||
|
Alcohol Ingredients Market Research FAQs
How big is the Alcohol Ingredients Market?
The Alcohol Ingredients Market size is expected to reach USD 8.39 billion in 2024 and grow at a CAGR of 1.75% to reach USD 9.16 billion by 2029.
What is the current Alcohol Ingredients Market size?
In 2024, the Alcohol Ingredients Market size is expected to reach USD 8.39 billion.
Who are the key players in Alcohol Ingredients Market?
Kerry Group plc, Cargill, Incorporated, Archer Daniels Midland Company, Dohler Group SE and Koninklijke Dsm N.V are the major companies operating in the Alcohol Ingredients Market.
Which is the fastest growing region in Alcohol Ingredients Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Alcohol Ingredients Market?
In 2024, the Europe accounts for the largest market share in Alcohol Ingredients Market.
What years does this Alcohol Ingredients Market cover, and what was the market size in 2023?
In 2023, the Alcohol Ingredients Market size was estimated at USD 8.25 billion. The report covers the Alcohol Ingredients Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Alcohol Ingredients Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Ingredients in Alcohol Industry Report
Statistics for the 2024 Ingredients in Alcohol market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Ingredients in Alcohol analysis includes a market forecast outlook to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
