Failure Analysis Market Size
| Study Period | 2019 - 2029 |
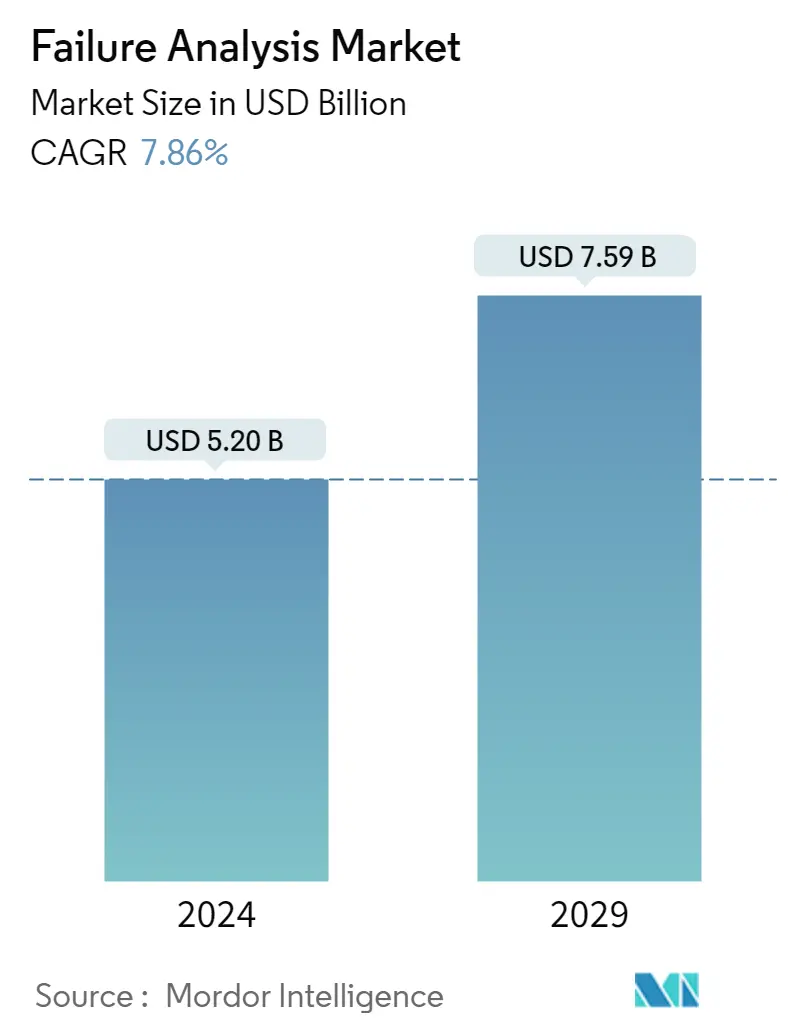
| Market Size (2024) | USD 5.20 Billion |
| Market Size (2029) | USD 7.59 Billion |
| CAGR (2024 - 2029) | 7.86 % |
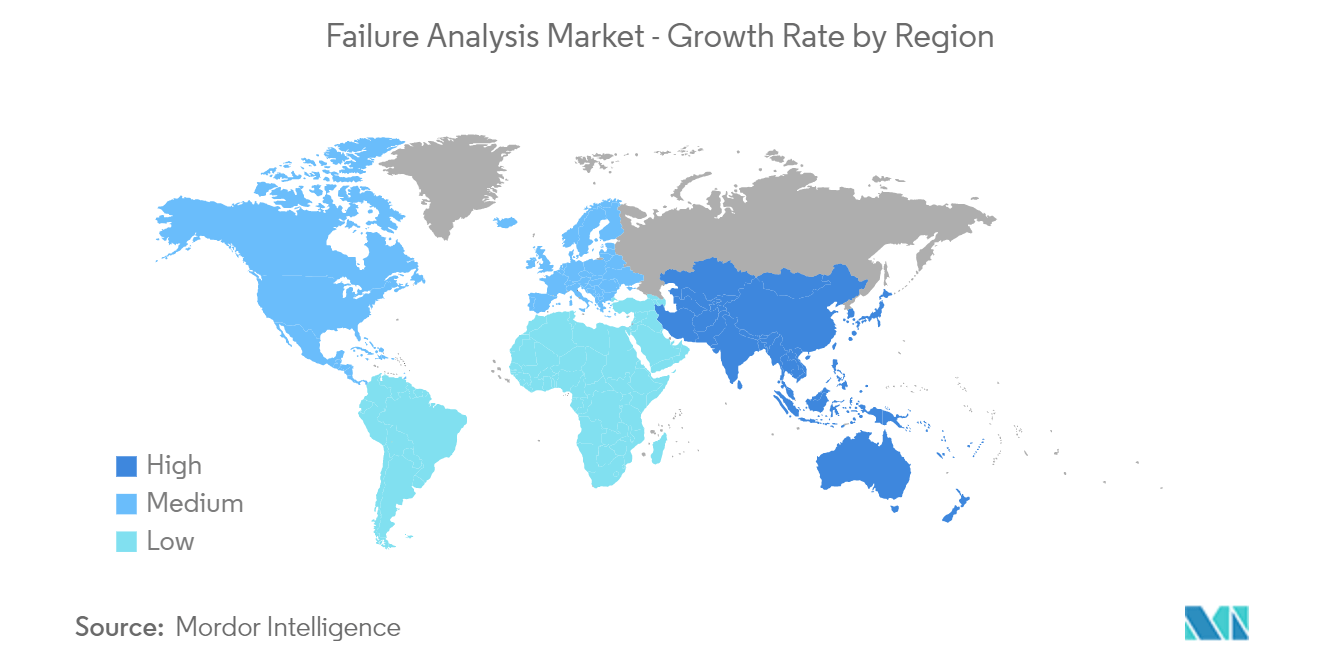
| Fastest Growing Market | Asia-Pacific |
| Largest Market | North America |
Major Players
*Disclaimer: Major Players sorted in no particular order |
Need a report that reflects how COVID-19 has impacted this market and its growth?
Failure Analysis Market Analysis
The Failure Analysis Market size is estimated at USD 5.20 billion in 2024, and is expected to reach USD 7.59 billion by 2029, growing at a CAGR of 7.86% during the forecast period (2024-2029).
The increasing requirement for failure analysis among automobile manufacturers, who also employ vision and artificial intelligence (AI)-based analysis tools for performance analysis, dynamic vehicle studies, accident reconstruction, etc., is augmenting the market's growth.
- Failure analysis refers to gathering and analyzing data to determine the cause of failure and corrective actions or liability. It is crucial to understand the root cause of failure to prevent similar incidents in the future. Common causes include assembly error, misuse or abuse, fastener failure, inadequate maintenance, manufacturing defects, low-quality material, improper heat treatments, unforeseen operating conditions, design errors, insufficient quality assurance, inadequate environmental protection, and casting discontinuities.
- Automation across the end-user industries has fueled the development in the market studied. Companies developing batteries for electric vehicles, mobile phones, energy systems, and other systems rely on testing to enhance the output, creating a more efficient, cleaner, and safer energy source. Companies use electron microscopy technologies to understand structures that level down at the atomic scale. They also use spectroscopy tools to discover critical changes in materials that cause defects and inefficiency.
- For the identification of problems and deficiencies in existing equipment, as well as those encountered with recent equipment or devices employed across different industries, failure analysis techniques have been applied. The demand for failure analysis equipment is increasing due to stricter use specifications, government safety rules, and high-quality control requirements. Regulatory authorities in various countries have jurisdiction over the development, production, and publication of technical industrial standards. For instance, the American Society for Nondescriptive Testing (ASNT) is a member that provides NDT-related reference materials, NDT training documentation, and technical conferences.
- The usage patterns of the microscopes have changed. Due to its advanced features, high resolution, and magnification power, conventional microscopes are gradually losing their market share in comparison with higher-end products like electron microscopes, scanning probe microscopes, and digital or hybrid microscopes. However, these microscopes are expensive, and this is a major factor limiting the use of them in hospitals, pathology laboratories, or small businesses.
Failure Analysis Market Trends
Automotive Sector is Driving the Demand for Failure Analysis
- The automotive industry has constantly faced new challenges as the demand for inexpensive, high-quality components has repeatedly increased. Extreme diligence must be imparted to ensure that such components are designed and manufactured with sufficient quality to withstand various service environments.
- Safety issues and product recalls, which may cost the company financially as well as its brand image, can be caused by defective products within the automobile industry. Visual defect detection allows manufacturers to find and classify defects in the parts of vehicles, like scratches, dents, or cracks, at a very accurate level. This helps avoid entry into production lines of faulty parts, improve quality, and increase safety and legal compliance.
- The automotive industry has witnessed rapid advancements in ADAS systems, connected vehicles, IoT devices, and smart sensors, which could prevent potential accidents due to malfunctioning of some of the vehicle's vital parts. According to a report published by the WHO in June 2022, every year, the lives of approximately 1.3 million people are cut short as a result of a road traffic crash. Between 20 and 50 million more people suffer non-fatal injuries, with many incurring a disability as a result of their injury.
- Potential causes for automotive device failure may include product design, manufacturing processes, contamination of the source materials, product packaging, improper handling, and improper calibration. These factors have raised concerns over the quality of manufacturing automotive parts and the preventive measures that can be implemented to avoid such incidents.
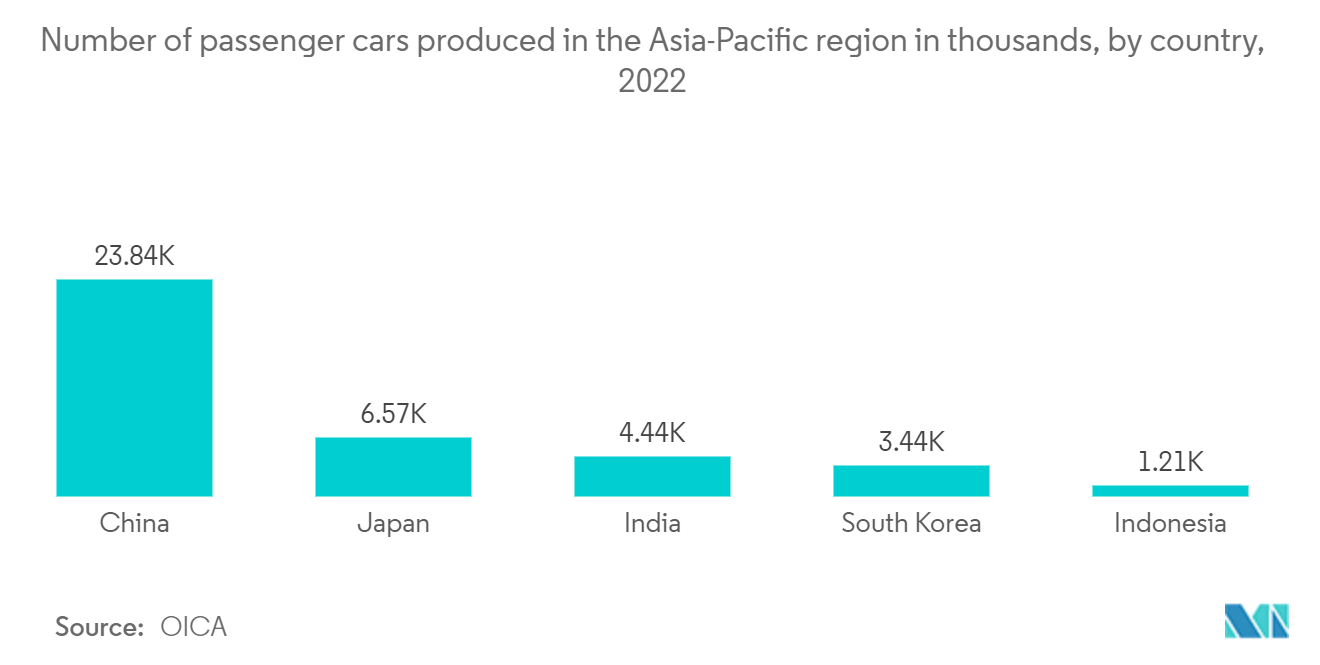
- Recently, the Asia-Pacific witnessed considerable growth in the automotive industry due to the increasing demand for passenger and advanced vehicles. Passenger vehicles find increased usage due to population density in the region. Therefore, the growing production of passenger and luxury cars is driving the market's growth in the region. According to OICA, China's automotive industry produced approximately 23.8 million passenger cars in 2022.
- The automobile component may fail in different failure modes, causing severe accidents. Therefore, it is essential to identify the failure mode for which the automobile component fails more frequently. Failure analysis is also done to establish the cause of an accident. The analysis is employed to see which component in the automobile caused the accident.
North America Occupies the Largest Market Share
- North America, majorly due to the United States, is one of the earliest adopters of failure analysis techniques, introduced originally to ensure no significant disruptions in the existing business models. The region is home to most of the prominent organizations in the world. For instance, the automotive sector in the country is ruled by General Motors and Ford, which rely heavily on automation.
- Furthermore, the United States is one of the leaders in crude oil production and has several oil rigs spread out across North America. The need for failure analysis techniques is being felt more acutely after the devastating effects of various oil rig accidents, the most notable being the Deepwater Horizon oil spill that occurred in 2010, discharging over 4.5 million barrels of oil into the sea due to a mechanical failure.
- The country has been witnessing critical infrastructure failures for the last few years. Two recent examples, an intense heat wave that pushed California's power grid to its limits in September 2022 and the failure of the water system in Jackson, Mississippi, amid flooding in August, show how a growing maintenance backlog and increasing climate change are turning the 2020s and 2030s into a golden age of infrastructure failure.
- To overhaul the US infrastructure, in November 2021, the Bipartisan Infrastructure Law (Infrastructure Investment and Jobs Act) was passed by Congress and signed into law by President Biden, seeking to invest significant investments in critical infrastructures. It emphasized key sectors to modernize the country’s infrastructure landscape, including improving and rebuilding roads, public transit, bridges, ports, rail, and airports.
- Similarly, to attract both private and public sector investment, the Canada Infrastructure Bank is responsible for investing USD 35 billion in revenue-producing infrastructure projects in Canada or parts of Canada by virtue of its investments.
Failure Analysis Industry Overview
The failure analysis market is highly competitive, featuring several key players. These leading companies, known for their significant market presence, are actively seeking to expand their operations across various industry sectors. Many of these firms are employing strategic collaborations to bolster their market share and profitability. Moreover, companies operating within this market are also engaging in acquisitions to enhance their product capabilities. Notable players in this sector include Exponent Inc., Rood Microtec GmbH, and EAG Inc.
In December 2022, Rood Microtec N.V. and NOVELDA proudly announced their partnership for the qualification, testing, and supply of NOVELDA's next-generation UWB (UltraWideband) devices. These UWB sensors have the remarkable capability of detecting even the slightest movements, as well as monitoring breathing and heartbeats. This groundbreaking technology positions NOVELDA as a provider of the most dependable human presence sensors worldwide. These sensors hold tremendous potential for various indoor applications, including consumer electronics, home automation, and smart buildings. The NOVELDA UWB Sensor promises to revolutionize user experiences, enhance safety, and contribute to energy conservation.
In May 2022, TEC Eurolab entered into a Memorandum of Understanding (MoU) with TWI, which became fully operational in October 2022. This MoU is expected to provide enhanced support to TWI members globally, with a particular emphasis on Europe and other cross-border collaboration opportunities. The agreement opens up opportunities for joint testing programs in various areas such as aeronautics, automotive, environmental testing, metal and non-metallic materials qualification, failure analysis, non-destructive testing, and more. This collaboration promises to advance the capabilities and services available in these fields.
Failure Analysis Market Leaders
Exponent Inc.
TEC Materials Testing
EAG Inc.
Rood Microtec GmbH
Presto Engineering Inc.
*Disclaimer: Major Players sorted in no particular order
Failure Analysis Market News
- April 2023: In order to cope with the increasing complexity and diversity of SiP Miniaturized Products' demand, USI developed Advanced Electronic Component Failure Analysis (FA) technology to respond to these new processes, materials, or packaging applications. USI uses laser layer removal and re-ball techniques for extraction of the main chip in the SiP module to mitigate process issues by using a high-resolution 3D x-ray that locates the position of abnormal components. In parallel, in order to identify the source of organic contaminants on the surface of the component, USI uses X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared spectroscopy (FTIR) to continuously improve its ability to analyze SiP module failure.
- January 2022: AEM's Afore Wafer-Level Test Solutions engineering team planned to install and exhibit the AIOLOS Wafer-Level Test Handlers at the RoodMicrotec facility in Nördlingen. This installation was part of a collaboration between RoodMicrotec NV, a leading independent company for semiconductor supply and quality services, and AEM, a global leader in semiconductor test and handling.
- January 2022: Mo., EAG Laboratories extended its medical device testing capabilities with a new 20,000-square-foot facility in St. Louis. The new laboratory was built specifically to meet the needs of the medical device sector. It aimed to improve EAG's analytical capabilities, provide faster turnaround times, and aid in meeting growing FDA and European Union's Medical Device Regulations criteria (MDR).
Failure Analysis Market Report - Table of Contents
1. INTRODUCTION
1.1 Study Assumptions And Market Definition
1.2 Scope Of The Study
2. RESEARCH METHODOLOGY
3. EXECUTIVE SUMMARY
4. MARKET INSIGHTS
4.1 Market Overview
4.2 Industry Attractiveness - Porter's Five Forces Analysis
4.2.1 Threat of New Entrants
4.2.2 Bargaining Power of Buyers
4.2.3 Bargaining Power of Suppliers
4.2.4 Threat of Substitute Products
4.2.5 Intensity of Competitive Rivalry
4.3 Industry Value Chain Analysis
4.4 Impacts of Macro Trends on the Market
5. MARKET DYNAMICS
5.1 Market Drivers
5.1.1 Ageing Infrastructure and Increasing Need for Maintenance
5.1.2 Technological Advancements Drive the Market Growth
5.1.3 Innovation in Materials, Design, and Production Methods
5.2 Market Restraints
5.2.1 High Equipment Cost Challenge the Market
6. TECHNOLOGY SNAPSHOTS ( TECHNIQUE)
6.1 Failure Modes Effect Analysis (FMEA)
6.1.1 Failure Modes, Effects, and Criticality Analysis (FMECA)
6.1.2 Functional Failure Analysis
6.1.3 Destructive Physical Analysis
6.1.4 Physics of Failure Analysis
6.1.5 Fault Tree Analysis(FTA)
6.1.6 Other Failure Mode Effect Analysis
7. MARKET SEGMENTATION
7.1 By Technology
7.1.1 Secondary ION Mass Spectrometry (SIMS)
7.1.2 Energy Dispersive X-ray Spectroscopy (EDX)
7.1.3 Chemical Mechanical Planarization (CMP)
7.1.4 Other Technology Types
7.2 By Equipment
7.2.1 Scanning Electron Microscope (SEM)
7.2.2 Focused Ion Beam (FIB) System
7.2.3 Transmission Electron Microscope (TEM)
7.2.4 Dual Beam System
7.3 By End-user Vertical
7.3.1 Automotive
7.3.2 Oil and Gas
7.3.3 Defense
7.3.4 Construction
7.3.5 Manufacturing
7.3.6 Other End-user Verticals
7.4 By Geography
7.4.1 North America
7.4.2 Europe
7.4.3 Asia-Pacific
7.4.4 Latin America
7.4.5 Middle East and Africa
8. COMPETITIVE LANDSCAPE
8.1 Company Profiles
8.1.1 Presto Engineering Inc.
8.1.2 TV SD Limited
8.1.3 Rood Microtec GmbH
8.1.4 EAG Inc.
8.1.5 MASER Engineering BV
8.1.6 NanoScope Services Ltd
8.1.7 CoreTest Technologies
8.1.8 TEC Materials Testing
8.1.9 McDowell Owens Engineering Inc.
8.1.10 Leonard C Quick & Associates Inc.
8.1.11 Crane Engineering
8.1.12 Exponent Inc.
- *List Not Exhaustive
9. INVESTMENT OUTLOOK
10. FUTURE OF THE MARKET
Failure Analysis Industry Segmentation
Failure analysis is an investigation of failure, which generally aims at identifying the cause and taking appropriate measures to correct it or mitigate its consequences. For the prevention of future asset and product failures, as well as to mitigate potential dangerous hazards for humans and the environment, failure analysis is carried out at all parts of the industrial sector, such as oil and gas, aerospace & defense, engineering & fabrication, power, marine, rail, automotive, electronics & sensors, medical equipment, etc.
The failure analysis market is segmented by technology (secondary ion mass spectrometry (sims), energy dispersive x-ray spectroscopy (EDX), chemical mechanical planarization (CMP), and other technology types), equipment (scanning electron microscope (sem), focused ion beam (FIB) system, transmission electron microscope (tem), and dual beam system), end-user vertical (automotive, oil and gas, defense, construction, manufacturing, and other end-user verticals), and geography (North America, Latin America, Europe, Asia-Pacific, and Middle East and Africa).
The market sizes and forecasts are provided in terms of value (USD) for all the above segments.
| By Technology | |
| Secondary ION Mass Spectrometry (SIMS) | |
| Energy Dispersive X-ray Spectroscopy (EDX) | |
| Chemical Mechanical Planarization (CMP) | |
| Other Technology Types |
| By Equipment | |
| Scanning Electron Microscope (SEM) | |
| Focused Ion Beam (FIB) System | |
| Transmission Electron Microscope (TEM) | |
| Dual Beam System |
| By End-user Vertical | |
| Automotive | |
| Oil and Gas | |
| Defense | |
| Construction | |
| Manufacturing | |
| Other End-user Verticals |
| By Geography | |
| North America | |
| Europe | |
| Asia-Pacific | |
| Latin America | |
| Middle East and Africa |
Failure Analysis Market Research Faqs
How big is the Failure Analysis Market?
The Failure Analysis Market size is expected to reach USD 5.20 billion in 2024 and grow at a CAGR of 7.86% to reach USD 7.59 billion by 2029.
What is the current Failure Analysis Market size?
In 2024, the Failure Analysis Market size is expected to reach USD 5.20 billion.
Who are the key players in Failure Analysis Market?
Exponent Inc., TEC Materials Testing, EAG Inc., Rood Microtec GmbH and Presto Engineering Inc. are the major companies operating in the Failure Analysis Market.
Which is the fastest growing region in Failure Analysis Market?
Asia-Pacific is estimated to grow at the highest CAGR over the forecast period (2024-2029).
Which region has the biggest share in Failure Analysis Market?
In 2024, the North America accounts for the largest market share in Failure Analysis Market.
What years does this Failure Analysis Market cover, and what was the market size in 2023?
In 2023, the Failure Analysis Market size was estimated at USD 4.82 billion. The report covers the Failure Analysis Market historical market size for years: 2019, 2020, 2021, 2022 and 2023. The report also forecasts the Failure Analysis Market size for years: 2024, 2025, 2026, 2027, 2028 and 2029.
Failure Analysis Industry Report
Statistics for the 2024 Failure Analysis market share, size and revenue growth rate, created by Mordor Intelligence™ Industry Reports. Failure Analysis analysis includes a market forecast outlook to for 2024 to 2029 and historical overview. Get a sample of this industry analysis as a free report PDF download.
